7: Precipitation Processes
- Page ID
- 9574
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Hydrometeors are liquid and ice particles that form in the atmosphere. Hydrometeor sizes range from small cloud droplets and ice crystals to large hailstones. Precipitation occurs when hydrometeors are large and heavy enough to fall to the Earth’s surface. Virga occurs when hydrometeors fall from a cloud, but evaporate before reaching the Earth’s surface.
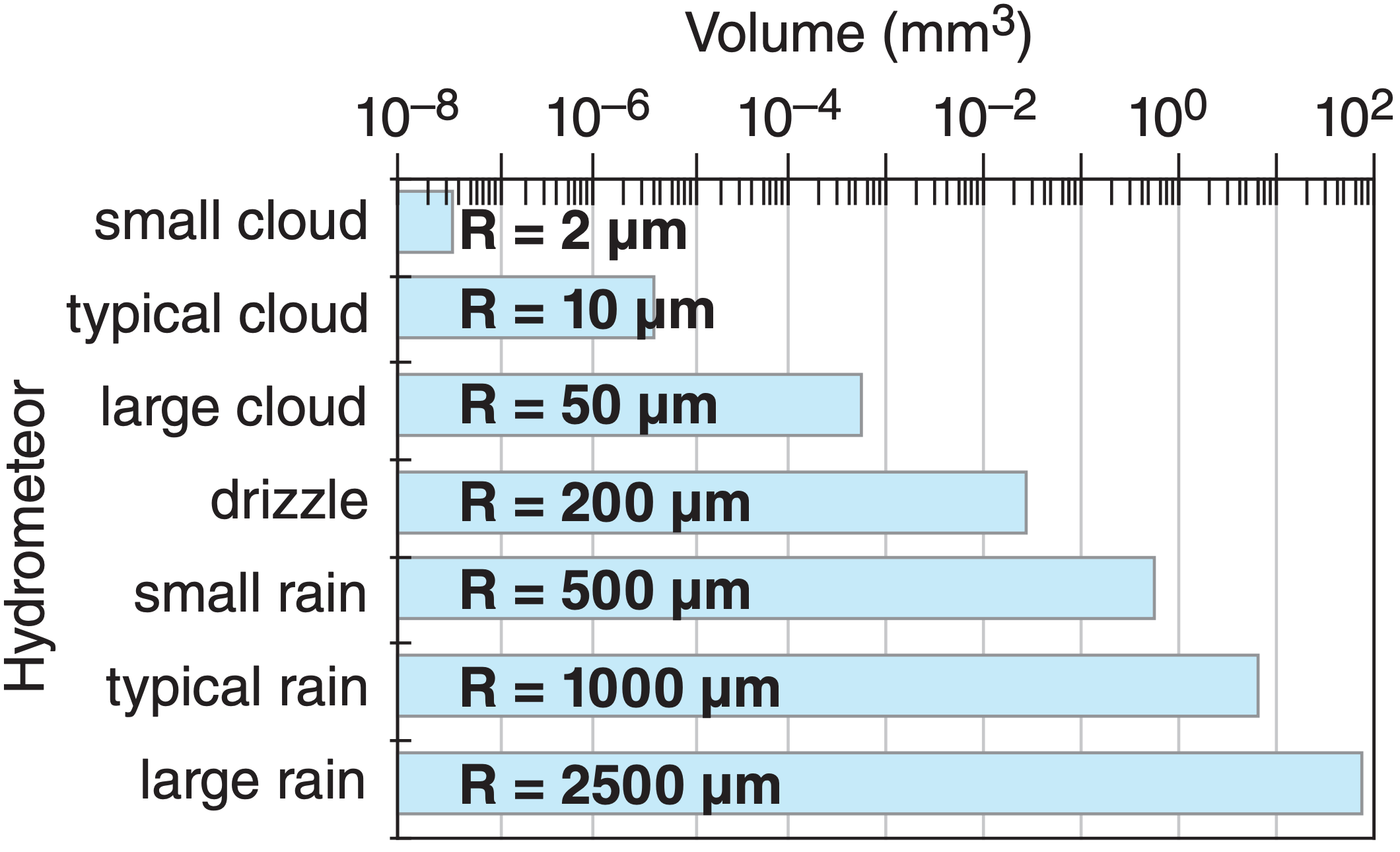
Precipitation particles are much larger than cloud particles, as illustrated in Fig. 7.1. One “typical” raindrop holds as much water as a million “typical” cloud droplets. How do such large precipitation particles form?
The microphysics of cloud- and precipitation-particle formation is affected by super-saturation, nucleation, diffusion, and collision.
Supersaturation indicates the amount of excess water vapor available to form rain and snow.
Nucleation is the formation of new liquid or solid hydrometeors as water vapor attaches to tiny dust particles carried in the air. These particles are called cloud condensation nuclei or ice nuclei.
Diffusion is the somewhat random migration of water-vapor molecules through the air toward existing hydrometeors. Conduction of heat away from the droplet is also necessary to compensate the latent heating of condensation or deposition.
Collision between two hydrometeors allows them to combine into larger particles. These processes affect liquid water and ice differently.
- 7.0: Homework Exercises
- This page focuses on enhancing understanding of atmospheric processes through research tasks and problem-solving in meteorology, including droplet and ice crystal formation, air pollution, and precipitation measurement. It presents quantitative exercises on cloud dynamics, terminal velocities, and the influence of cloud condensation nuclei (CCN) on precipitation.
- 7.1: Supersaturation and Water Availability
- This page covers two main topics: supersaturation in the atmosphere, which occurs when air holds more water vapor than its equilibrium state, and the relevant calculations for supersaturation fractions using vapor pressures. It explains the significance of water availability for hydrometeor formation and their growth limitations before precipitation. Additionally, it discusses cloud droplet sizes, specifically 21.2 µm and 9.
- 7.2: Nucleation of Liquid Droplets
- This page covers nucleation processes for cloud droplets, focusing on homogeneous vs. heterogeneous nucleation, emphasizing the role of cloud condensation nuclei (CCN). It explains the Köhler equation, detailing the impact of curvature and solute on droplet growth, and discusses stable vs. unstable equilibria in droplet formation illustrated by the Köhler curve.
- 7.3: Nucleation of Ice Crystals
- This page explains the conditions for ice formation, emphasizing that ice forms below 0°C under normal pressures. It contrasts homogeneous freezing, which occurs in supercooled droplets near –40°C, with heterogeneous freezing that involves impurities. Four processes of heterogeneous freezing—deposition, immersion, condensation, and contact—are described, each with specific mechanisms.
- 7.4: Liquid Droplet Growth by Diffusion
- This page explains the growth of droplets in a supersaturated environment, emphasizing the role of humidity gradients and diffusion. It describes how larger droplets create gentler gradients, affecting moisture flux and the growth rate of smaller droplets. Additionally, the page presents calculations for droplet radius over time under 1% supersaturation, detailing parameters like pressure and temperature.
- 7.5: Ice Growth by Diffusion
- This page covers the formation and characteristics of ice crystals in the troposphere, highlighting the hexagonal shape and growth processes under varying conditions. It explains the Wegener-Bergeron-Findeisen process, illustrating how ice crystals develop in supercooled liquid water clouds. Key factors such as temperature, supersaturation, and ice nuclei concentration are discussed, emphasizing their roles in precipitation formation.
- 7.6: Collision and Collection
- This page explains precipitation formation in both warm and cold clouds. Larger hydrometeors fall faster due to greater mass, enhancing growth and precipitation potential through collisions and mergers. It details warm-cloud processes, including droplet coalescence, and cold-cloud processes that involve aggregation of ice crystals.
- 7.7: Precipitation Characteristics
- This page delves into rain-drop size distributions and categorizes rainfall intensity into light, moderate, and heavy, using the Marshall-Palmer distribution as a model. It provides tables for precipitation intensity, drizzle attributes, and blizzard conditions. World-record rainfall events illustrate the significance of intensity-duration-frequency relationships for infrastructure.
- 7.8: Precipitation Measurement
- This page provides an overview of instruments for measuring precipitation, such as manual and automated rain gauges, disdrometers, and snow depth sensors. It details traditional methods like tipping-bucket and weighing gauges, alongside advanced techniques involving light beam attenuation and particle imaging. It also discusses liquid-water content probes, ultrasonic sensors, and remote sensing technologies, including weather radar and satellites, noting their applications and accuracy.
- 7.9: Review
- This page explains cloud droplet formation and growth, highlighting the role of condensation nuclei and surface tension. In warm clouds, fewer CCN lead to larger droplets and increased rainfall through collision and coalescence. In colder clouds, ice nuclei promote crystal growth, which collects moisture from liquid droplets. This process can quickly lead to graupel or hail. Most midlatitude rain results from melted snow due to these cold cloud processes.
Thumbnail: https://www.pexels.com/photo/selecti...-rain-1463530/


