13.4: Algal Blooms
- Page ID
- 45627
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Intense dinoflagellate blooms occur naturally, but infrequently, in many parts of the coastal oceans, particularly in subtropical to temperate climates. The frequency of such blooms has been observed to be increasing, and they are occurring in areas where they have not been seen before.
Dinoflagellate blooms can be caused by a variety of mechanisms. For example, a large increase in freshwater runoff can form a shallow surface layer of low-salinity water in a previously well-mixed water column. Phytoplankton that were previously light-limited because they were frequently mixed below the photic zone may be restricted to this shallow surface layer, where they are always in the photic zone. This situation may trigger a bloom. In some instances, it is thought that runoff containing dissolved inorganic or organic compounds may trigger blooms because these substances react with toxic substances, such as copper or mercury, and reduce their toxicity to the phytoplankton. Many blooms are caused by such natural events, but increasing nutrient fertilization due to human activity (Chap. 16) is generally believed to be a significant causative factor in the increasing frequency and severity of blooms.
Because the nitrogen compound concentration in discharges of treated sewage wastewater and of agricultural runoff and industrial waste are high in relation to the silicate concentration, such discharges are generally believed to be responsible for the higher frequency of dinoflagellate blooms, as compared to diatom blooms. However, several cyanobacteria, also called “blue-green algae”, convert dissolved molecular nitrogen, a nitrogen form that most phytoplankton cannot use, to usable nitrate. In some areas, these cyanobacteria may provide the nitrogen needed to initiate or sustain dinoflagellate blooms.
Dinoflagellate blooms can appear quickly but often disappear within a few days as nutrients become limiting. However, under appropriate conditions, blooms can last for many weeks if the source of nutrients is sustained. Dinoflagellates reproduce very rapidly. In optimal conditions, dinoflagellate populations can double in as little as a few hours. Accordingly, blooms can grow within a few days to concentrations of dinoflagellate cells a million or more times greater than normal. Dinoflagellates can also concentrate themselves into blooms by using their weak swimming ability to congregate at depths where light levels are optimal.
The dense concentration of phytoplankton in blooms colors the surface water yellow, green, brown, or reddish, depending on the species. Many dinoflagellates are reddish in color, so their blooms are often called “red tides.”
Dinoflagellate blooms often have adverse effects on other marine organisms and, occasionally, on human health. These effects are caused by toxic substances synthesized by certain phytoplankton species, particularly when they are stressed in bloom situations, or by the depletion of oxygen caused by the decaying organic matter that remains when the bloom collapses.
Dinoflagellate and Other Plankton Toxins
Like many terrestrial plants, certain phytoplankton species produce toxic substances as a defense against herbivores. Some, but not all, species of dinoflagellates produce particularly virulent toxins, including saxitoxin, brevetoxin, okadaic acid, and domoic acid. These substances are complex organic compounds and often mixtures of several compounds, some of which are many times more toxic than, for example, strychnine. The toxins are selective: some are toxic to vertebrates only, whereas others also affect some invertebrates, particularly crustaceans such as shrimp and crabs. Toxins produced by dinoflagellate blooms often kill large numbers of fish.
In addition to fish kills, dinoflagellate toxins can cause serious human health problems because the toxicity is selective for vertebrates. Most filter-feeding organisms (organisms that strain or sift food particles from the water), including clams, mussels, and oysters, are unaffected by dinoflagellate toxins but concentrate the toxins in their body tissues. Other invertebrates, such as crabs that feed on filter-feeding invertebrates, can also become contaminated with these toxins from their food without being affected. If any of these contaminated invertebrates and shellfish are eaten by vertebrates, the toxins can still cause their deadly effects in vertebrates. Unfortunately, humans are one of the vertebrate species that feed on shellfish and other invertebrates.
Human poisoning by dinoflagellate toxins concentrated in shellfish is well known. At least four major pathologies are caused by shellfish-borne toxins. The most widespread is paralytic shellfish poisoning. The toxins that cause paralytic shellfish poisoning are strong nerve poisons that can cause permanent nerve injuries or, if the concentrations are high enough, paralysis and death. There are an estimated 1600 annual cases of paralytic shellfish poisoning worldwide, and an estimated 300 fatalities among these cases. Neurotoxic shellfish poisoning is less common and less serious than paralytic shellfish poisoning, but it produces symptoms that are often mistaken for the more common types of bacterial food poisoning. Diarrhetic shellfish poisoning is also less serious but probably more widespread than neurotoxic shellfish poisoning, and it causes severe diarrhea. Amnesic shellfish poisoning is a much rarer pathology, in which short-term memory is destroyed, perhaps permanently.
These toxins can also affect coastal communities when winds blow ashore sea spray containing dinoflagellate cells or their detritus from nearshore blooms. When this happens, the toxins cause the local human population to suffer from allergy-like reactions, such as irritated eyes, running noses, coughs, and sneezes.
Most dinoflagellate toxins are stable compounds that remain toxic even after prolonged cooking of contaminated shellfish. Once the bloom has disappeared, some shellfish lose their toxicity within days, but other species may remain toxic for months. The only certain way to avoid human poisoning by these toxins is to identify affected shellfish and prevent them from reaching the marketplace. Many countries, including the U.S., have extensive seafood monitoring programs to ensure toxin-free shellfish. Unfortunately, these monitoring programs are extremely expensive and difficult to implement. As an alternative, in areas where dinoflagellate blooms occur with reasonable frequency, shellfishing areas are closed to harvesting during the summer and fall, when blooms are most likely. Such closures lead to substantial loss of income for fishers. Areas closed to shellfishing because of dinoflagellate blooms are expanding each year, perhaps indicating an increase in the frequency and geographic extent of such blooms. For example, while historically infrequent, blooms of the dinoflagellate Karenia brevis now recur annually. These blooms typically develop in late summer or fall off Florida’s coast. They mainly occur off the Gulf Coast, but can also be found off the Atlantic coast.
At one time, only dinoflagellates were thought to produce the powerful toxins that affect people and other vertebrates. However, blooms of at least two different diatom species have been observed to produce domoic acid. An event that occurred in 1987 in Prince Edward Island, Canada, left several victims afflicted with amnesic shellfish poisoning. The victims suffered short-term memory loss that lasted at least 5 years. A second event, in 1991 off the west coast of California, Oregon, and Washington, did not affect people. In this case, pelicans were poisoned by domoic acid produced by a diatom species that had been consumed by anchovies, on which the pelicans were feeding. Shellfisheries throughout this huge area were closed to harvesting for many weeks.
Humans and pelicans are not the only vertebrates poisoned by phytoplankton toxins. For example, in 1986, hundreds of bottlenose dolphins died off the coasts of New Jersey and Maryland after consuming fish contaminated with a dinoflagellate toxin, and more died in the same way off the Carolina coast in 1987. In these cases, the fish were either less susceptible to the toxin than the dolphins, or the dolphins accumulated more of the toxin as they fed on large quantities and were unable to destroy or excrete the toxin.
Other groups of planktonic organisms also produce toxins. For example, cyanobacteria produce toxins that have caused numerous fish kills in freshwater. In 2010, a toxin from cyanobacteria was found to be responsible for the deaths of sea otters. This event was the first known occurrence of hepatotoxic shellfish poisoning in the marine environment. The cyanotoxins apparently originated in freshwater rivers but were consumed by coastal shellfish, which were, in turn, consumed by the sea otters. It has been hypothesized that these toxins may be produced by bacteria or viruses living in the phytoplankton cells, rather than by the phytoplankton themselves. More recently, a persistent bloom of the diatom Pseudo-nitzschia off the Southern California coast in 2023–2025 produced domoic acid, a neurotoxin that killed hundreds of sea lions, dolphins, and whales.
Harmful algal blooms (HABs) now occur in the waters of almost every coastal state of the U.S. and are estimated to be responsible for direct economic losses of between $10 million and $100 million annually. Costs are incurred in public health, the closure of commercial fisheries, and fisheries management and monitoring. The most frequent toxic algal blooms occur off the Atlantic coast of Florida every year. It has been estimated that the impacts of these blooms alone cost tens of millions of dollars annually, and a 2018 red tide event in Florida is estimated to have an estimated total economic impact of $2.7 billion. Algal blooms in other areas have caused deaths of many species, including pelicans, sea otters, and bottlenose dolphins, and tainted commercially valuable species, causing closures of recreational and commercial fisheries and major economic losses. In 2015, one of the largest and most severe HABs occurred along the entire West Coast of North America from southern California to the Aleutian Islands. The principal organism in the bloom was a diatom that produced domoic acid, but there were dinoflagellates that produced toxins that cause paralytic and diarrhetic shellfish poisoning in some parts of the affected area. The bloom lasted from May until October and resulted in the closure of several important fisheries. Closures imposed an estimated $48 million in economic losses for the Dungeness crab fisheries, which remained closed until March 2016, $22 million in the Pacific razor clam fishery, and major losses in other closed shellfisheries. The bloom toxins were also implicated in the deaths of many top predators, especially sea lions, and several seabird species.
Oxygen Depletion and Dead Zones
Below the permanent thermocline, oxygen concentrations are reduced by oxygen consumption during respiration and decomposition of detritus that has been transported through the thermocline from the photic zone (Chap. 12). The same processes consume oxygen below seasonal thermoclines, but the reduction in oxygen concentration is usually small for two reasons. First, the residence time of water in the layer below a seasonal thermocline (but above any permanent thermocline) is at most a few months. When the seasonal thermocline breaks down in the fall, oxygen is returned to this water when it becomes part of the mixed layer in contact with the atmosphere. In addition, in locations where a seasonal thermocline forms, the total production of phytoplankton during the summer is limited by nutrient availability in the shallow mixed layer above the seasonal thermocline. Hence, only a limited quantity of organic matter is transported through the thermocline to be decomposed. Areas with high productivity are generally areas of upwelling or other physical processes that prevent the formation of a steep thermocline. Vertical mixing in these areas continuously transports oxygen-deficient water to the surface, where it regains oxygen from the atmosphere.
Severe oxygen depletion does not generally occur below seasonal thermoclines, but it may occur in specific circumstances when the oxygen demand in the subthermocline layer is very large. The cumulative oxygen demand is the total amount of oxygen consumed before the water mass recontacts the atmosphere or before additional oxygen is supplied by mixing with water that has a higher oxygen concentration. Two circumstances tend to produce high cumulative oxygen demand: high productivity in the overlying water, and long residence time of water below the thermocline. If oxygen concentrations become too low, many animal species cannot survive for more than a short period of time. This low oxygen condition is called hypoxia. If the oxygen concentration drops to zero—a condition called anoxia—animals cannot survive at all.
Residence times of water below seasonal thermoclines on the continental shelf are generally short, but residence times of water below permanent thermoclines in marginal seas or ocean inlets and bays may be very long (often many decades). Consequently, despite the relatively low productivity of marginal seas that have steep thermoclines, the cumulative oxygen demand is sufficient in certain locations to deplete oxygen. The result can be anoxia and the formation and accumulation of toxic sulfide in bottom waters. Examples of such locations are many Norwegian and other fjords and in the Baltic Sea, where anoxic conditions have been present continuously in the deep waters of the central region for at least 100 years. The geographic area of anoxic bottom waters in the Baltic has grown during that time in response to increasing inputs of nutrients and oxygen-demanding organic matter by humans (Chap. 16).
In some marginal seas or other coastal areas, normal conditions result in periodic (often seasonal) or permanent hypoxia. In some of these regions, increased phytoplankton growth and blooms can occur as a result of nutrient inputs from sewage, agriculture, and other human sources. Blooms produce large amounts of detritus, and as a result, the naturally low oxygen concentrations below the thermocline are reduced further. This may lead to occurrences of anoxia, causing most benthic biota and some deep-water nekton to be either killed or deprived of these areas as habitat.
Anoxia may occur in any particular year in a coastal region if primary productivity is high and/or residence time is long. For example, on the continental shelf of New York and New Jersey in 1976, wind mixing was limited, and an intense bloom of phytoplankton developed. The combination of long residence time and high productivity caused widespread anoxia that extended for hundreds of miles along the coast in a band tens of miles wide across the shelf (Fig. 13-15a). The anoxia killed enormous numbers of many marine species, including clams and other shellfish valued at more than $500 million.
The 1976 anoxia was not caused directly by excess nutrients discharged to the oceans from New York and New Jersey, because natural conditions were responsible for the unusually long residence time of that year. However, nutrient inputs to this region in sewage increase productivity and, thus, the probability of blooms. If nutrient inputs are allowed to increase, hypoxia and anoxia would occur at shorter residence times and, hence, probably more frequently.
Overfertilization of natural waters with nutrients that cause blooms and sometimes hypoxia or anoxia is called eutrophication. It has been well known in lakes for decades, but has only more recently become the subject of intense study in the coastal oceans.
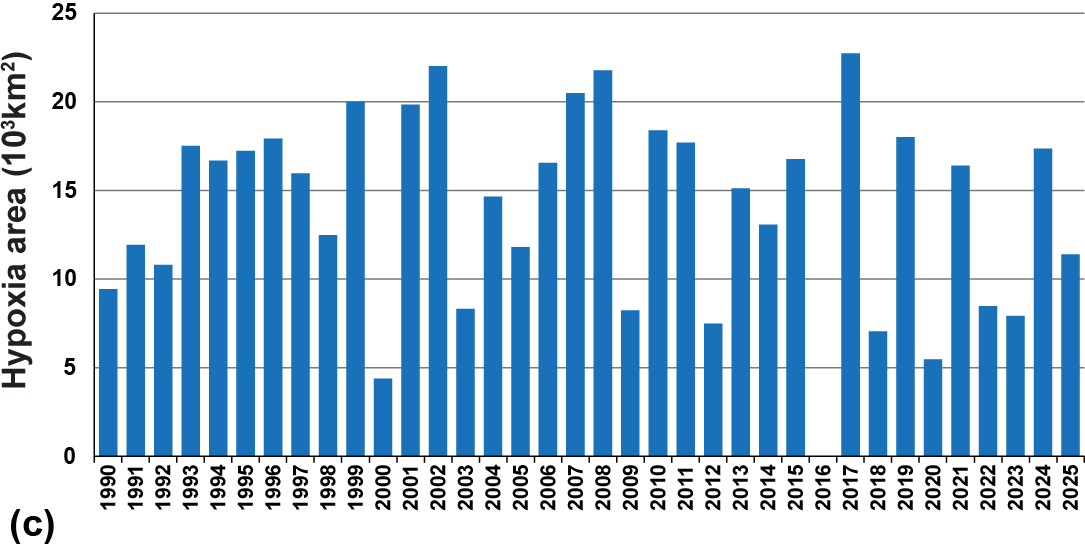
Temporary or permanent hypoxia or anoxia has been found in a growing number of areas of the coastal zone, increasing from just a handful of locations in the mid-20th century to hundreds today and still increasing. This problem is now so widespread that a new term “dead zone” has become a common way to describe such occurrences. For example, in the Adriatic Sea, the Saronikos Gulf in Greece, the Sea of Japan, and many bays and harbors periodic massive algal blooms exceed the consumption capacity of the zooplankton population and cause anoxia in the bottom waters. The blooms have been so bad in some years in the Adriatic Sea that the rotting algae have fouled beaches and the air, severely affecting tourism in Italy and Croatia. In the U.S., hypoxia or anoxia has occurred in the North Atlantic coastal zone, the Gulf of Mexico, Chesapeake Bay, the Oregon coastal region and a number of other bays and estuaries. Extensive efforts have been made to reduce nutrient inputs to Chesapeake Bay to lessen the severity of anoxia, but progress has been disappointing so far. In the Gulf of Mexico, there is a seasonal dead zone that now develops annually and often extends from near the mouth of the Mississippi River to the Mexican border of Texas (Fig. 13-15b,c).
The anoxia that now occurs annually during the summer on the Oregon continental shelf is apparently caused by a new mechanism not previously seen. The mid-depth water mass in the Pacific Ocean has an oxygen minimum zone due to the prolonged time this water mass has been below the thermocline. This oxygen minimum zone appears to be expanding and becoming thicker since it is now shallow enough to extend onto the Oregon continental shelf when winds create seasonal upwelling in summer. The cold high-nutrient water that is upwelled normally supports abundant marine life in this region. However, the upwelled water now often contains insufficient oxygen for respiration and causes a dead zone with massive kills of marine animals. The apparent expansion and deepening of the oxygen minimum layer at mid depths in the North Pacific may be a reflection of either a slowing of the MOC circulation (Chap. 8) or an increase in ocean primary production due to increased temperatures and nutrient availability in ocean surface waters, or perhaps both. If the expansion continues, more coastal upwelling regions will likely experience periodic hypoxia, and the area of the oceans that has a mid-depth water mass that has too little oxygen to support animal life will expand.