9.4: Evolutionary Patterns in Fossils
- Page ID
- 33306
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Figured Stones to Fossils: Nicolas Steno
Fossils are evidence of ancient life. However, they were not always seen as such. The evolution of our understanding of these figures in stone is a classic example of how science progresses with new knowledge. Since the time of the ancient Greeks, people have noticed seashells in stones far from shore and well above current sea level. Greek writers including Xenophanes, Herodotus, Strabo, and Eratosthenes interpreted them being left behind by the ebb and flow of the sea. They also viewed the Earth as eternal. Perhaps because later Christian thinkers saw this view of the Earth as too Pagan, in conflict with the Biblical account in Genesis, these views eventually fell away from acceptance in Western Europe. Still, people wondered about the odd shapes in local rocks, referred to at the time as “figured stones”. Today, we know them as fossils.
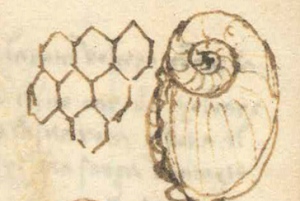
They saw them contained within the building blocks of cathedrals, along pathways, and in the mountains. Fossils of marine shellfish, long pulled from the limestones of the Alps, were not seen as evidence of former life despite their similar appearance with living shellfish of the time. Or if they were originally from the sea, the preferred interpretation was that they were a remnant of the global deluge, a result of the deposition of organisms along a globally flooded, and eventually inundated land surface. Others interpreted the figures in the rocks not as evidence of flood deposits but as having been placed there during creation itself. Divinely created as is, they were considered to be miraculous. Still the ultimate nature of fossils remained elusive to most, until Nicolas Steno (1638-1686), a Danish scientist, had another idea.
Steno was trained as an anatomist. He also had many other interests. Cutler (2003) recounts Steno’s life, beginning with his famed skill at dissection. These skills led him to many discoveries, including the salivary glands in mammals. This fame led him to the Medici court in Florence, Italy, where he not only found financial support, but also was given the opportunity to study anything he wanted. This freedom of curiosity and creativity would lead eventually to his geological explorations. It would be these geological discoveries, rather than anatomical insights, that would make Steno famous as the “father” of geology.
Steno was curious about the “figured stones”. Some contained what looked like seashells while still others contained structures that the local people called “tongue stones” (Figure \(\PageIndex{2}\)). After studying in detail the head of a shark specimen reeled in by two fishermen in 1666, Steno would make the connection between the similar shape of that shark’s teeth and the “tongue stones” found encased in rocks nearby. He was the first to publish accurate interpretations regarding how such a “solid within a solid” could be possible, suggesting that these tongue stones represented evidence of ancient life: tongue stones were the teeth of long-dead sharks! He argued that the encased solid (the shark tooth in this case) had to first exist on its own, prior to being encased. This line of reasoning is an early form of his “principle of relative dating by inclusions”, that is still utilized today. His recognition of these “figured stones” as fossils would lay the groundwork for a rebirth of thought about Earth’s past that would carry into the minds of enlightenment thinkers just decades later. As scientific understandings of observation and experimentation would advance, so too would our appreciation of fossils as evidence of the history of life itself, including our species’ own backstory.

Other scientists of Steno’s time would fully accept fossils as being evidence of ancient life. One such contemporary was Robert Hooke (1635-1703), who studied fossils under the microscope. While conducting this work, he noticed similarities between features in certain fossils and extant organisms. He noted that mineralization could occur between wood fibers, producing permineralized wood. He also determined that shells could be turned into fossils after being filled with mud or clay. After Steno, two key individuals would emerge whose work and ideas would lay the groundwork for the use of fossils toward understanding past life.
The first was Carl von Linné (a.k.a. Carolus Linnaeus; 1707-1778). Linneaus was interested in fossils, and while he did not recognize them yet as evidence of prior life, he did acknowledge they were distinctively different than other kinds of rocks and minerals. Linnaeus’ original classification system included three kingdoms. These were Regnum Animale (Animal), Regnum Vegetabile (Vegetable), and Regnum Lapideum (Mineral). While these labels are still used today, our current classification schemes for animals, plants (vegetable), and geological materials (rocks and minerals) are very different. In Linnaeus’ Mineral Kingdom, he did take pains to do what others before him struggled with. He separated fossils out as being different from rocks. He did not call them evidence of ancient life, but he recognized that they were different in some way.
The second important paleontologist of this time was Georges Cuvier (1769-1832). He fully accepted fossils as being evidence of ancient life. Because Cuvier saw fossils as ancient life, he was the first to include them in the Linnaean system along with living forms. During his life he collected a massive amount of fossil material from the New World. Using this collection, he would eventually argue for the existence of extinction through his study of megafaunal fossils, in particular. The extinction of an entire species was not thought to be possible, or at least common, in nature. Cuvier’s revolutionary ideas surrounding extinction, supported by the fossil record, represent a paradigm shift in how fossils were viewed. Not only were they evidence of ancient life, but they were also evidence of wide-scale ancient death. And, extinctions make way for evolutionary change.
The Principle of Faunal Succession
Because the fossil record shows evolution occurring through time, we can reconstruct the story of life. Cladistic analysis, the discovery of transitional fossils, and the abundance and variety of organisms that have been discovered all support the evolutionary story of descent with modification. Because of this, when we find a rock with a particular fossil in it, we can assign a relative age to it because we have a good understanding of the succession, or sequence, of organisms in the record. Biologist J.B.S. Haldane (purportedly in a conversation with Paul Harvey in 1970), once asked how his confidence in the theory of evolution could be challenged, replied, “fossil rabbits in the Precambrian”. There is little concern that such an outlandish discovery would, however, negate evolutionary theory. Rather, such a radical change in our understanding of fossil succession would merely cause scientists to re-evaluate timelines, originations of species, and other associated evidence toward a recasting of the theory in the light of such new knowledge.

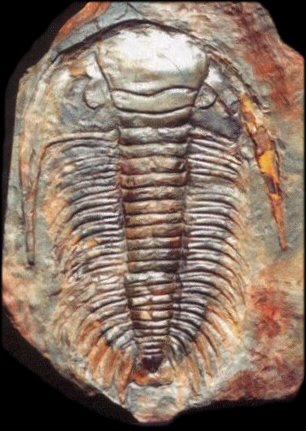
One excellent example of how succession works is with trilobites. These classic and important fossils among invertebrates were a key part of the “Cambrian Fauna” (Sepkoski, 1984). Originating most abundantly in the Cambrian and truly flourishing through the Ordovician Period, they went extinct at the end of the Permian Period, victims of the largest mass extinction in Earth’s history. Because of this, we find no examples of trilobites prior to their origination during the latest Precambrian and none after the start of the Triassic. Knowing the time of their origination and the time of their extinction and the ability to reconstruct the clades within this fossil group’s phylogeny provides a variety of key index fossils that can be used to identify rocks of particular ages, all because scientists have been able to reconstruct their evolutionary history. For instance, Paradoxides pinus is a trilobite typical of the middle Cambrian while the Isotelus maximus is characteristic of the middle to late Ordovician. The presence of these specimens in a rock is key evidence that the host rocks were deposited during these periods of time.
An Inherently Biased Record: Signor-Lipps Effect
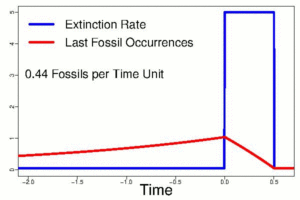
Evolution is well supported in the fossil record. Even so, the limitations of this record create significant challenges for understanding the history of extinct life. For instance, using cladistics to organize known fossils into clades based upon shared and derived characteristics is very useful. However, only a small percentage of life is fortunate enough to come down to us as a fossil, allowing its story to be told. Significant gaps, or missing data, occur in the record. This bias in the record tends to be against taxa with no hard skeletal parts, those that are generally fragile, and those that live in habitats where rapid burial is unlikely or other circumstances are not ideal for fossilization. Within the time range of a species, there are great uncertainties about its origination and its extinction, when it appears and when it dies off. In fact, these exact moments in the life of a species are entirely impossible to determine. The cause of this effect is a sampling bias, related to a variety of causes, but most critically to the overall problem that not everything gets fossilized and, what has been fossilized is not all that has been found. Philip Signor and Jere Lipps (1982) described this sampling bias, now referred to as the Signor-Lipps Effect, because the oldest appearances of a given taxon appear to come on gradually just as the youngest appear to die away gradually, even if the extinction (or origination) itself was actually quite sudden.
The Cretaceous/Paleogene extinction event that ended the reign of reptiles is an excellent case study for this challenge (Alvarez et al., 1980; Signor and Lipps, 1982). Until Walter and Luis Alvarez identified an asteroid as a cause of this geologically sudden extinction, mass extinctions were thought to come on quite gradually because of the apparently slow disappearance of fauna in uppermost Cretaceous strata. The Signor-Lipps Effect signal in the rock record below the extinction boundary suggested that it was gradual – a matter of relative appearance. Their work showed otherwise and so proved that such events can happen suddenly, despite what the record of fossils might appear to be showing.
Transitional Fossils
Transitional fossils are fossilized remains that share ancestral characteristics and those of the derived group of descendants. By this definition, and considering how rapid cladogenesis can occur, there can be a wide interpretation of exactly what constitutes a transitional fossil. Some workers would say that all fossils are transitional fossils! Generally the idea is invoked only between major groups. Table 1 illustrates a wide variety of these, but the classics that come to mind most rapidly for most are Archaeopteryx sp., an organism that is transitional between non-avian dinosaurs and birds (Dengler, 2018), Pakicetus sp., a transitional organism between land mammals and whales (Gingerich et al., 2018), and Tiktaalik, an organism that shares the characteristics of lobe-finned fish while also displaying the derived characters seen in later terrestrial vertebrates (Carroll, 2016).
|
Fossil Group or Clade |
Organism |
Features |
Image |
Between: |
Period |
|---|---|---|---|---|---|
|
Avialae |
Archaeopteryx lithographica |
Jaws with sharp teeth, Long bony tail, Feathers |
|
Non-Avian Dinosaurs and Birds |
Late Jurassic (150 Ma) |
|
Stegocephalia |
Tiktaalik roseae |
Scales, Gills, Flattened head, Sturdy interior fin bones, Ears capable of hearing in both air and water |
|
Fish and Amphibians |
Late Devonian (375 Ma) |
|
Hominin |
Ardipithecus ramidus |
Grasping big toe, Reduced canine tooth size, bipedal features in the pelvis and base of skull |
|
Common ancestor of chimpanzees and humans |
Late Miocene (4.4 Ma) |
|
Epicynodontia |
Thrinaxodon liorhinus |
Possibly had hair, secondary palate, Nasals pitted with foramina, inner ear bone |
|
Synapsids to Mammals |
Early Triassic (250 Ma) |
|
Cetacea |
Basilosaurus cetoides |
First fully aquatic mammal, nearly complete loss of hindlimbs |
|
Land Mammals to Whales |
Late Eocene |
Paleobiogeography
One of the major factors in fossilization that is important to consider when contemplating the record is that not all organisms lived in every environment and everywhere around the world. As is true of most organisms today, even humans, geographic distribution of organisms is determined by the environments to which they have adapted. There have been fossils, such as the Glossopteris sp. ferns and Paradoxides sp. trilobites that have been found in myriad locations, apparently far apart from one another. These far flung fossil sites have helped reconstruct paleogeography (positions of continents) because of the abundance of specimens and similarity of environment.

Because of our understanding of modern ecology and evolutionary principles, we can go beyond just describing fossils and organizing them by appearance, we can also apply biogeographic understandings to place them within ecosystems and, sometimes, even describe communities within those ecosystems. At times, there are very interesting stories that emerge when examining the paleobiogeography of an organism or group, when epiboles (sudden appearances) or outages (sudden disappearances) occur, giving us insights into the stability and resilience of long-extinct organisms and environments.
For further exploration of paleobiogeographic material, the Paleobiobology Database Navigator is an amazing tool.
Homology, Analogy, and Vestigiality
Humans are tetrapods, a trait we share with all four-legged organisms going back to the earliest amphibians and fish. Such homology, or similarity in structures, supports cladistic analysis. Organisms with similar developmental biology, or embryology, will tend to develop the same structures. However, through evolutionary change, or descent with modification, these same structures can be adapted for different uses. Structures within a lineage, such as the arm bone sequence within tetrapods (one upper arm bone, two lower arm bones, many hand bones), are considered to be homologous structures. These shared characteristics, derived from a much earlier ancestor, are key identifiers within a lineage. For our own species, the arm bone sequence is a classic case that connects us to the earliest tetrapod, Tiktaalik rosaceae, the earliest freshwater organism to carry this trait onto land that we know of from the fossil record. Homology provides critical insights into how heredity works. Thus, as Pierre Belon noticed in 1555, humans and birds have similar structures in their arms. Birds being adapted to flight, their wings are otherwise useless for the manipulation of tools, climbing trees, or some of the other uses for which a primate descendent might use them. Homology implies divergent evolution, or the splitting off of lineages through the processes of speciation, because advantageous traits develop in different ways for different organisms.

Not all similar structures are homologous. Some structures are analogous. Wings are an excellent example, for while birds have wings as mentioned above, so do butterflies. The reason bird and butterfly wings are analogous is that these structures evolved independently of one another. These analogous structures are examples of convergent evolution, or the independent evolution of similar structures among different taxa that, can also share a similar adaptational purpose, such as wings assisting with flight in the case or birds and butterflies. There are many other examples of convergent evolution and analogy. Consider the similar torpedo-shaped swimming forms of tuna, icthyosaurs, dolphins, and penguins. Some of the best examples occur among plants, such as the over 60 independent instances of C4 photosynthesis, one of several photosynthetic pathways for producing sugars, that evolved among 19 different families of plants over time (Osborne and Sack, 2012), wind-blown seed dispersal, and carnivory.
Finally, it is worth considering vestigial structures. These are anatomical features, cladistically shared with ancestral taxa, that serve little to no function purpose or can even be maladaptive. Such structures can be hard to identify but are not uncommon. They are the result of a lack of positive selective pressure, meaning that the structure no longer provides a function that leads to an evolutionary advantage. These structures, sometimes also referred to as vestigial organs, can be found in an atrophied, or reduced form, in many instances. A classic example is the loss of sight in blind cave fish. Living a life in total darkness, these fish no longer have need to expend energy maintaining a sensory organ evolved for light detection. While they retain eyes, they are no longer functional. Other examples of organisms retaining structures that are mere vestiges of their past usefulness are wings in some island-dwelling birds, such as penguins or Galapagos cormorants, or hindlimbs that are retained in whales and snakes.
Care must be taken in labeling structures definitively as vestigial for the reason of exaptation (Gould, 1982), a kind of re-adaptation of an evolved structure for a different purpose. Feathers are an example of a feature that could have become vestigial but instead was the subject of exaptation. Having originally been evolved to aid in thermal management, feathers eventually became just as important for purposes of display.
Pan troglodytes right forelimb by Blackburn Lab on Sketchfab
Origination as an Evolutionary Mechanism
Origination is speciation. It is the process by which new species evolve from ancestral forms. As this evolution occurs, phylogenies grow. Though often visually depicting a sense of gradual change, phylogenies are anything but gradual in the way they develop. They branch out as they develop over time, but off-shooting branches tend to occur pretty suddenly in the fossil record. Speciation, which ultimately creates new groups, does not occur at a constant rate over time. Rather, it is most typically quite choppy. Much like a real tree, the tree of life has branches leading out from a trunk and smaller branches popping out quite suddenly. To analyze evolutionary relationships among groups, Huxley (1959) introduced the concept of a clade, or group of related organisms, including a common ancestor and all its related descendants. Cladistic analysis provides insights into relationships between organisms through character analysis–studying the observable features of organisms in order to establish whether they are shared or derived from an ancestral taxon. Shared characters are traits that evolved early in a clade and are visible among the entire group. All primates, humans included, have opposing thumbs, for example. These are also called synapomorphies. Derived characters are traits that distinguish a taxon within its clade. They are also called apomorphies. While shared traits define an entire clade, derived traits represent evolutionary innovation that leads to the formation of new clades through speciation. Distinguishing these traits among a population of organisms or fossils is often done using a character matrix, where traits of taxa are listed one one axis and the tax themselves on the other.

Figure \(\PageIndex{9}\): A character matrix can be used to analyze evolutionary relationships among organisms. Shared derived characters, rather than shared ancestral traits, are used to parsimoniously differentiate between a group of organisms. (Image: University of California Museum of Palentology, https://evolution.berkeley.edu/admin/media)

Figure \(\PageIndex{10}\): From a character table, a cladogram can be created to visually explore shared derived characters, their development through speciation (origination), and their development over time. In this case, connections between sharks, primates and other mammals, and dinosaurs are explored in a larger scale. Red bars indicate the development of a shared derived trait and subsequent speciation events that led to the development of additional clades over time. (Image: University of California Museum of Palentology, https://evolution.berkeley.edu/admin/media)
Such character analysis makes it possible to create cladograms (Figure \(\PageIndex{10}\)) by organising taxa first by shared characters and then through the identification of derived characters over time.The cladograms used to represent evolutionary relationships very intentionally imply a sense that most of the time, life is enjoying a kind of stasis but that, every once in a while, is punctuated by a major event, called species origination, that creates a new branch off of the tree. This more modern view of speciation, or cladogenesis, as the generation of a new clade through speciation is called punctuated equilibrium (Gould and Niles Eldredge, 1972). This set of ideas relies on the idea that environments retain a sense of stability unless changed, such as through major environmental upheavals. Punctuated equilibrium predicts that groups of organisms will otherwise remain in periods of stasis with little evolutionary change through most of their existence. Cladogenesis is, then, a rather geologically sudden process.
Within a cladogram, groups of organisms are ordered according to time across the horizontal axis, and evolutionary change along the vertical axis. There are three ways organisms can be grouped in a cladogram, based upon how they are related to one another. A monophyletic group is a clade, or a group of organisms that all share common traits derived from a common ancestor and all of its descendants. Such groups include not only the ancestral organism from which all shared characters are derived, every organism included across time and evolutionary change beyond that. Paraphyletic groups are not defined as clades. In contrast with monophyletic groups, these groupings of organisms retain common characteristics among related organisms, but not all of the descendent groups of organisms. The lemurs, lorises, and tarsiers below are examples, but so are reptiles, with regard to other amniota. While they contain a great many of amniotic organisms within their paraphyletic group, they do not contain the mammals or birds, which are also amniotes. The final situation is that which results from convergent evolution, or homoplasy. Polyphyletic groups are situations where organisms may have evolved similar characteristics but these characters are not derived from a common ancestor. In the case of lorises and tarsiers, both groups are nocturnal, a behavioral trait that is not shared with or derived from any other primates and must have evolved independently of one another. Another example would be mammals and birds, who are both haemothermic, or warm-blooded, a characteristic not derived from a common ancestor but also evolved through homoplasy.

Extinction as an Evolutionary Mechanism
Extinction is the opposite of origination. It is the permanent termination of an organism or a group of organisms. At this moment in time, 99% of all species that have ever existed have been subject to extinction. In actual numbers, about 5 billion species are thought to have succumbed to such a demise. The lifespan of some species is certainly longer than others, and geographic distributions also vary. Because of these two factors, it can be very challenging to determine the actual moment of extinction. Nevertheless, extinction is very common and perfectly normal. At any given moment in the geologic past, species are going extinct. It is the rate of extinction that varies. Background extinction rates are much different from the rate of extinction during mass extinction events.
Normal background extinction rates vary because species life spans vary. Estimates for the life spans of various taxa range from a high of 11 million years to as short as 0.5 million years for all fossil groups (Lawton and May, 1995). This means that some species, not individual organisms of course, could be around for as long as 11 million years. Our own species, Homo sapiens, has been around for about the last 300,000 years (0.3 million years). Comparing this span of time to the low end average estimate, it could be said that we have a long way to go before our species becomes extinct. But, such a numerical comparison does not necessitate such a reality!

In terms of background extinction rates, estimates hover between 0.1 genera per million genera year for fossil taxon and 1 extinction per million species per year for living taxa. This translates to about 10-100 species per year going extinct, globally. The fossil record estimates are coarse, due to preservation bias (favorite marine organisms with mineralized skeletons) and inherent destruction in the record over time (the near-present is better represented than the ancient past). Modern estimates are difficult also due to the fact that so many species are not yet known and because the challenge of taking species census data is quite large. The modern numbers, when parsed more simply, suggest that for birds we would expect one extinction every 400 years or one mammal every 200 years. These estimates take into account the lifespan of species. Mammals, as a group, are thought to have an average species lifespan of about 1 million years.
When extinction rates increase sharply above background rates for a geologically short period of time, the event is referred to as a mass extinction. Mass extinctions are rare, but when they happen they make huge adjustments to the patterns of life on the planet. Traditionally, there five really large mass extinctions that are set aside as special. In order, they mark the end of the Ordovician Period, the end of the Devonian Period, the end of the Permian Period, the end of the Triassic Period, and the end of the Cretaceous Period. Very recently, a sixth ancient mass extinction is being discussed that would directly precede the End-Permian event. Called the end-Guadalupian extinction, it would have occurred just 8 million years prior to the traditional end-Permian event. Ultimately, Raup and Sepkoski (1982) first identified the “Big Five”.
- background extinction - the normal, ongoing rate of species loss that occurs as a result of natural ecological and evolutionary processes
- homology - a similarity in structure, genetics, or developmental pathways among different organisms that results from shared ancestry
- mass extinction - a short time interval during which an exceptionally high proportion of Earth’s species becomes extinct across multiple ecosystems and taxonomic groups
- vestigial structures - anatomical features or behaviors that persist in an organism but have lost most or all of their original function through evolution







