1: The Basics of Climate Change
- Page ID
- 34456
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Search Fundamentals of Biochemistry
(Learning Goals written by Claude, Anthropic)
By the end of this chapter, students should be able to:
The Greenhouse Effect and Earth's Energy Balance
- Explain how greenhouse gases — especially CO₂, CH₄, and N₂O — trap infrared radiation emitted by Earth, using IR spectroscopy and blackbody radiation principles to connect molecular properties to planetary warming, and calculate the Earth Energy Imbalance (~1 W/m²) to appreciate its magnitude.
- Define Global Warming Potential (GWP₁₀₀), compare the values, atmospheric lifetimes, and climate contributions of CO₂, CH₄, and N₂O, and explain why CO₂ remains the primary long-term concern despite methane's higher per-molecule potency.
- Rebut the argument that "CO₂ absorption is saturated" using three lines of evidence: spectral line broadening at higher pressures and Doppler effects, continued absorption in the upper troposphere and stratosphere, and direct satellite measurements showing decreased outgoing IR radiation at CO₂ absorption wavelengths.
Climate History: Ice Ages, Orbital Forcing, and CO₂
- Interpret ice core and ocean sediment proxy data to describe the synchronous relationship between atmospheric CO₂ and temperature over 800,000 years, explain what the Milankovitch cycles (eccentricity, obliquity, precession) do and do not explain, and identify why present CO₂ levels are unprecedented in at least 3 million years.
- Explain how small orbital temperature increases trigger deglaciation through AMOC slowdown, Southern Ocean warming, and CO₂ release from ocean stores — making CO₂ the driver of the bulk of subsequent warming — and rebut the skeptic argument that "temperature rises before CO₂."
- Describe the role of dust, iron deposition, and phytoplankton primary production as feedback mechanisms that amplify or dampen glacial cycles through their effects on ocean CO₂ uptake.
Deep-Time Climate: Key Events and Sensitivity
- Describe the Paleocene–Eocene Thermal Maximum (PETM, ~56 MYA) and the Eocene–Oligocene Transition (EOT, ~33 MYA) as case studies in rapid CO₂-driven climate change, comparing PETM CO₂ release rates (~1.5 Pg/year) to present emissions (~25 Pg/year).
- Distinguish equilibrium climate sensitivity (ECS, ~3.4°C per CO₂ doubling) from Earth System Sensitivity (ESS, ~5–8°C), explain what each includes, and connect the higher ESS value to the long-term consequences of sustained CO₂ elevation.
- Explain why AMOC weakening (projected ~50% by 2100) represents a major climate risk, describing its consequences for European weather, North American sea levels, tropical precipitation, ocean carbon uptake, and atmospheric CO₂.
Introduction
We've known for a long time that burning fossil fuels and releasing CO2 into the atmosphere would warm our climate. Perhaps the first paper addressing this, Circumstances affecting the Heat of the Sun's Rays, was published in 1856, before the US Civil War, by a woman scientist, Eunice Foote. John Tyndall (of the Tyndall effect) published more comprehensively on greenhouse gases in 1859. Given the complexity of the biosphere's climate, it was not until the 1980s that climate models became sophisticated enough for scientists like James Hansen to become convinced and alarmed enough to discuss, in Congressional hearings, the role of anthropogenic (human-caused) CO2 released into the atmosphere as a cause of ever-worsening global warming. The knowledge of human-induced climate change has been politicized and subjected to an orchestrated campaign of misinformation and disinformation by fossil fuel companies and their political contributors. We have delayed global action for so long that we must act immediately and aggressively to address climate change before we reach climatic conditions so austere for humans that parts of the world become uninhabitable. Homo sapiens evolved in a world dominated by repetitive glaciation and deglaciation. Hans Joachim Schellnhuber, an atmospheric physicist, climatologist, and founding director of the Potsdam Institute for Climate Impact Research, has stated that humans have so affected the world that we have eliminated the possibility of the next glaciation cycle.
Many readers might not be familiar with the data and models supporting human-caused climate change. They may not be aware that climate scientists are almost unanimous in supporting climate data and models. As in any field, however, you will find outliers who don't and whose ideas carry disproportionate weight among climate change skeptics. Hence, we provide basic data to show the relationships among increasing atmospheric CO2 levels, global warming, drought and flooding, ocean acidification, and biodiversity loss. We also provide supportive information, allowing users to address questions from those who question the reality of present human-induced climate change. We don't shy away from using basic physics either, since most students studying biochemistry at the level covered in this book have also studied physics and biology. In subsequent sections, we will address the biochemical consequences of climate change and how biochemistry can help mitigate it.
Greenhouse Effect
Before the advent of the Industrial Revolution, the Earth's climate was fairly constant since the last ice age, which peaked about 22,000 years ago (YA) and ended about 12,000 YA. There have been short (on a geological time scale) cooling periods since the end of the last ice age. Humans evolved around 200,000 years ago, with modern civilizations arising about 4000 BCE, so it could be said that humans are ice-age peoples (a distinctly Northern Hemisphere perspective). Humans have benefited from a fairly stable climate since then.
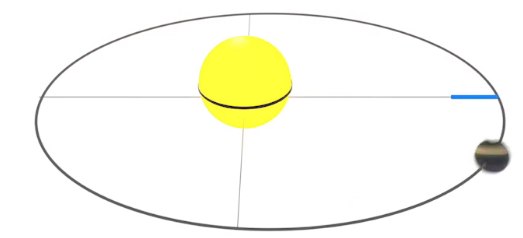
The sun's energy warms the Earth. If the Earth did not radiate back into space an amount of energy equal to what it receives, it would slowly and continually warm. The Earth reflects energy as light. In addition, as the Earth is heated by the sun, it emits infrared radiation (as do all warm objects). Earth's temperatures are stable when the energy it emits equals the energy it receives from the sun. This is illustrated in Figure \(\PageIndex{1}\).
Figure \(\PageIndex{1}\): Energy balance for Earth as it adsorbs and emits solar radiation
Relatively constant levels of atmospheric CO2 have enabled our stable climate, a trace atmospheric gas, which has hovered around 280 parts per million (ppm) until the start of the Industrial Revolution in 1770. CO2 is a greenhouse gas, which, as anyone who has run an IR spectrum knows, absorbs in the infrared. CO2 in the atmosphere absorbs some of the infrared radiation emitted by the Earth, allowing the Earth to be warmer than it would be in its absence. The CO2 effectively acts as an insulating blanket. In fact, without CO2 or other "greenhouse" gases, the Earth would be completely covered by snow.
Other greenhouse gases in the atmosphere include methane and nitrous oxide. The IR spectra of these gases are shown in Figure \(\PageIndex{2}\). Students who have taken organic chemistry labs and obtained IR spectra of samples always blank the instrument to remove spectral signals from both CO2 and H2O.
Figure \(\PageIndex{2}\): IR spectra of some greenhouse gases. NIST (ex: https://webbook.nist.gov/cgi/cbook.c...ndex=1#IR-SPEC)
Let's look at the relationship between the IR spectra of CO2 and the spectra emitted by the sun and the Earth. You have probably heard of a blackbody radiator in introductory physics classes. It is a hypothetical object that absorbs 100% of all electromagnetic radiation that falls on it without transmitting or reflecting it (hence it appears black). In addition, it emits light energy that depends only on its temperature, not on its chemical composition.
The surface of the sun is about 5778 K. The peak of blackbody radiation for a body at that temperature is at approximately 0.5 μm (500 nm), which falls in the middle of the visible spectrum. The Earth, with an average temperature of 288 K, emits at a peak of about 10-15 μM in the IR region. The blackbody radiation curves emitted by the sun and the Earth are shown in the Figure \(\PageIndex{3 }\) PHET simulation below. Move the blackbody temperature slider to see the effect on the emitted blackbody radiation. You will have to change the -/+ icons to see the Earth's IR spectra in comparison to the sun.
Figure \(\PageIndex{3}\): PHET Interactive Simulations. University of Colorado, Boulder. https://phet.colorado.edu/en/simulat...ectrum/credits
Play with the slider and magnifying glass icons to view the Earth's emission spectrum (peak at 8.3 μM, spanning 5-16 μM). This overlaps with the CO2 IR absorption peak (Figure 2) centered at 667 cm-1 (15 μM) with a wavelength range span of about 500–850 cm⁻¹ (12-20 μM). The sun's spectrum has little intensity at 10-15 μM, so it contributes nothing to CO2 absorbance at those wavelengths. It's light just passes through the atmosphere until it strikes Earth.
Since the start of the Industrial Revolution, humans have been releasing ever-increasing amounts of CO2 into the atmosphere from burning fossil fuels, methane from agricultural practices, and natural gas production. As of January 2025, CO2 levels have reached 427 ppm, while methane has increased to 1900 parts per billion (ppb) or 1.9 ppm.
Interactive graphs showing atmospheric CO2 and methane over time are shown in Figures \(\PageIndex{4}\) below.
Figures \(\PageIndex{4}\): Atmospheric CO2 and methane over the last 800,000 years. Our World in Data.
Increasing atmospheric methane contributes about 20% of the global warming effect of more concentrated CO2, as it has a strong IR absorption spectrum. It has a short atmospheric half-life (about 20 years) compared to CO2 (hundreds of years). Nitrous oxide (N2O) is also a powerful greenhouse gas that depletes stratospheric ozone. Our increased use of synthetic fertilizers and manure is the primary anthropogenic source of N2O. Its emissions are accelerated by poorly drained farmlands.
The net effect is that the Earth is no longer in energy balance. The net contributions of energy input and output are expressed as energy/sec/m2 of the Earth's surface area. Hence, the units are Joule/s.m2 = Watts/m2, where 1 Watt is 1 J/s. From around 2005 to 2019, the average Earth Energy Imbalance (EEI) (or budget) was about +460 x 1012 W or 460 terawatts (TW) = 4.6 x 1014 W. Dividing this by the surface area of the planet (5.1 x 1014 m2) gives an average global value of +0.90 ± 0.15 W/m2 (or J/(m2s)). Newer and more refined estimates show it to be +1 W/m2, with most of the excess energy stored in Earth's Oceans, which have a high specific heat capacity and cover 71% of Earth's surface. Most of the remaining excess energy is used to warm land masses and melt ice shields and glaciers. 1 W/m2 might not seem like a lot, but it is (pardon the pun), astronomically big.
Figures \(\PageIndex{5}\) below shows the general, continual increase in the ocean heat content (OHC) in zetaJoules (ZJ, 1021 J) of the upper 2000 m of the ocean since 1960, with a dramatic acceleration since 2000.
Figures \(\PageIndex{5}\): Global upper 2000 m OHC from 1958 through 2025 according to (a) IAP/CAS, (b) CIGAR-RT (from 1961 through 2025), and (c) Copernicus Marine (from 2005 through 2025) (1 ZJ = 1021 J). In panels (a) and (b), the black curves represent monthly time series, while the histograms show annual anomalies. Pan, Y., Cheng, L., Abraham, J. et al. Ocean Heat Content Sets Another Record in 2025. Adv. Atmos. Sci. (2026). https://doi.org/10.1007/s00376-026-5876-0. Creative Commons Attribution 4.0 International License. http://creativecommons.org/licenses/by/4.0/. Here is a link to the Heat Content in the top 700 meters of the ocean - Our World in Data
The atomic bomb dropped on Hiroshima released about 1.8 x 1013 Joules (some estimates are as high as 9 x 1013 Joules) of energy. Assume for the sake of calculation that this energy was released in 1 second over the planet's entire surface (5.1 x 1014 m2). The earth energy imbalance for that one explosion was 0.035 J/m2.s (0.176 J/m2.s for the higher estimate). The ratios of the present imbalance (+1 W/m2) to the distributed bomb's imbalances are 1/0.035 = 28x for the lower energy estimate bomb or 1/0.176 = 5.7 for the higher energy estimate. Hence, the present imbalance, 1 J/m2s from human-caused climate change, is equivalent to detonating 6-28 Hiroshima-type atomic bombs every second. Other estimates are lower and closer to 600 bombs per day. Regardless, these values should give you a dramatic sense of the imbalance and how unsustainable it is.
Figure \(\PageIndex{6}\) below represents the Earth's energy budget.
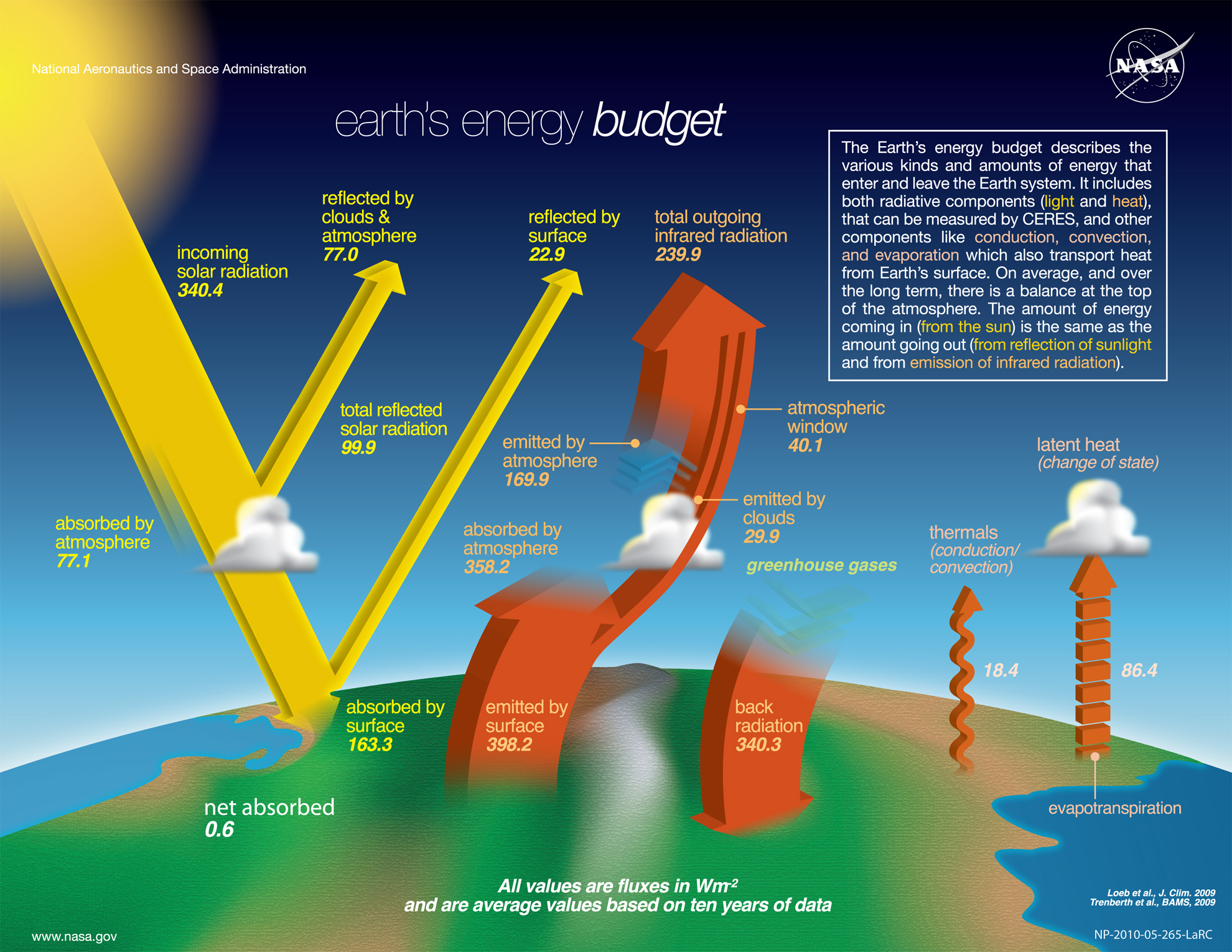
Figure \(\PageIndex{6}\): Earth's energy budget, with incoming and outgoing radiation (Values are shown in W/m2). Satellite instruments (CERES) measure the reflected solar and emitted infrared radiation fluxes. Year 2021 update: Net absorbed energy (shown as 0.6) rose to above 1.0 W/m2 based on independent CERES and ocean heat content measurements. (see Fig. 1 in Loeb et al.(2021), Geoph. Res. Let 48 (13), doi:10.1029/2021GL093047). Public Domain
The global warming potential (GWP) calculates the total contribution of all emitted greenhouse gases. It is expressed in units of CO2 equivalents. It accounts for the contributions of other greenhouse gases, such as CH4 and nitrous oxide (N2O), each with unique IR absorption spectra (as shown in Figure 2 above) and atmospheric half-lives. The IPCC uses a 100-year time frame for the calculation of the GWP, which is often abbreviated as GWP100, and uses this formula:
\begin{equation}
\mathrm{CO}_2 \text { equivalent } \mathrm{kg}=\mathrm{CO}_2 \mathrm{~kg}+\left(\mathrm{CH}_4 \mathrm{~kg} \times 28\right)+\left(\mathrm{N}_2 \mathrm{O} k g \times 265\right)
\end{equation}
- CO2 has a GWP of 1 by definition since it is the reference. Its time frame in the atmosphere (100s to 1000 years) doesn't matter since it is the reference.
- CH4 has a GWP of around 27-30 over 100 years, reflecting its higher IR absorbance but lower lifetime (around 12 years).
- N2O has a GWP of around 265-273 over a 100-year timescale and a lifetime of around 109 years.
Water is also a greenhouse gas, as you can attest on humid days, and its lack in the atmosphere over deserts leads to a large temperature drop at night. It's very different than other greenhouse gases. Its concentration varies enormously (from 40 ppm to 40000 or more) based on humidity and precipitation events, which remove it from the atmosphere. The amount of water in the atmosphere increases with rising global temperatures, leading to more intense precipitation events and further warming in a positive feedback loop. Its concentration in the atmosphere changes significantly on time scales of hours and days, so its atmospheric half-life is short. In contrast, the half-life of CO2 in the atmosphere is measured in decades to centuries.
The basis for methane's contribution to climate change needs a clearer explanation. Given its IR absorption spectra, it is a very potent greenhouse gas. Many oil companies have indicated a commitment to decreasing methane leakage from pipelines, which would be helpful. Still, these efforts may draw attention away from the fact that CO2 is the biggest problem. Over 20 years (not 100 as in the GWP calculations above), methane is 80 times as potent as CO2. Those numbers sound ominous (80X and 20 years, which is not long away)! But you shouldn't compare the two on a kg/kg basis, as the warming from CO2 could last 1000 years, whereas that from methane lasts only 20 years, after which it disappears. Given its short atmospheric lifetime, the long-term climate effect of methane depends on the rate at which it is added to the atmosphere (megatons/year). Still, for CO2, it is the net mass added over a longer time, which has a cumulative effect. CO2 effects continue to increase, but methane effects wane quickly. So you can't easily compare warming over the first 20 years. Consequently, a delay in acting on methane is minimal compared to a delay in acting on CO2.
Methane contributes to about 30% of net warming, and about 60% of the methane is probably from human activities. As we hit the IPPC Paris target of 1.5 0C, about 30% of that, or about 0.45 0C, is from methane; hence, 0.27 0C comes from human sources. Decreasing human sources of methane will help. But we can't use our efforts to remove methane to avoid or slow down the push for net-zero CO2 emissions.
Climate changes over the last million years
Climate has always changed. Our present period is no different, so action is unnecessary.
Indeed, the Earth has been subject to cycles of glaciation and deglaciation for hundreds of thousands of years. Luckily, we can determine atmospheric CO2 levels dating back hundreds of thousands of years by measuring the entrapped CO2 in ice cores from Antarctica and Greenland. In addition, we've been able to infer temperatures over this time frame using proxies (tree rings, fossils, and more, as described in section 31.2). Figure \(\PageIndex{7}\) shows how atmospheric CO2 and temperature have varied over the last 800000 years using Antarctic ice core data.
|
Figure \(\PageIndex{7}\): Atmospheric CO2 concentrations (ppm, red) and temperature deviations from the past 1000 years (green) using ice core data and temperature proxies. The bottom figure shows the same graph with the duration of the repeated ice ages shown in light blue. Antarctic Ice Cores Revised 800K YrCO2 Data. Bereiter, B.; Eggleston, S.; Schmitt, J.; Nehrbass-Ahles, C.; Stocker, T.F.; Fischer, H.; Kipfstuhl, S.; Chappellaz, J. http://ncdc.noaa.gov/paleo/study/17975
Several key features of the graph should be apparent:
- Both atmospheric CO2 and temperature change (ΔT) are periodic. So yes, it is true that "climate changes," as climate change skeptics argue.
- Both CO2 and ΔT change in synchrony. An obvious question is: what changes first? Does ΔT drive CO2 changes or vice versa? More on that in a bit.
- The CO2 levels in more recent times (on the right-hand side of the graph) have soared to levels not seen in the last 800,000 years! CO2 emissions from the burning of fossil fuels cause this change.
Figure \(\PageIndex{7 zoom}\) below zooms in to show both CO2 atm and temperature values (look at changes, not actual values) since 1000 CE. It also includes annotations on relevant climate and historical events.
Figure \(\PageIndex{7 zoom}\): CO2 atm (blue) and temperature values (red, look at changes, not actual values) since 1000 CE. Note that they generally proceed in lock step. Data and data sources from the 2 Degrees Institute.
Over those 800K years, the Earth has experienced cycles of glaciation/deglaciation, including recurring ice ages. Figure (\PageIndex{8}\) below shows a depiction of the last ice age, which peaked 21,000 years ago (left). At that time, the ice cap over New York City was about 1 mile high (right) as CO2 was 185 ppm!
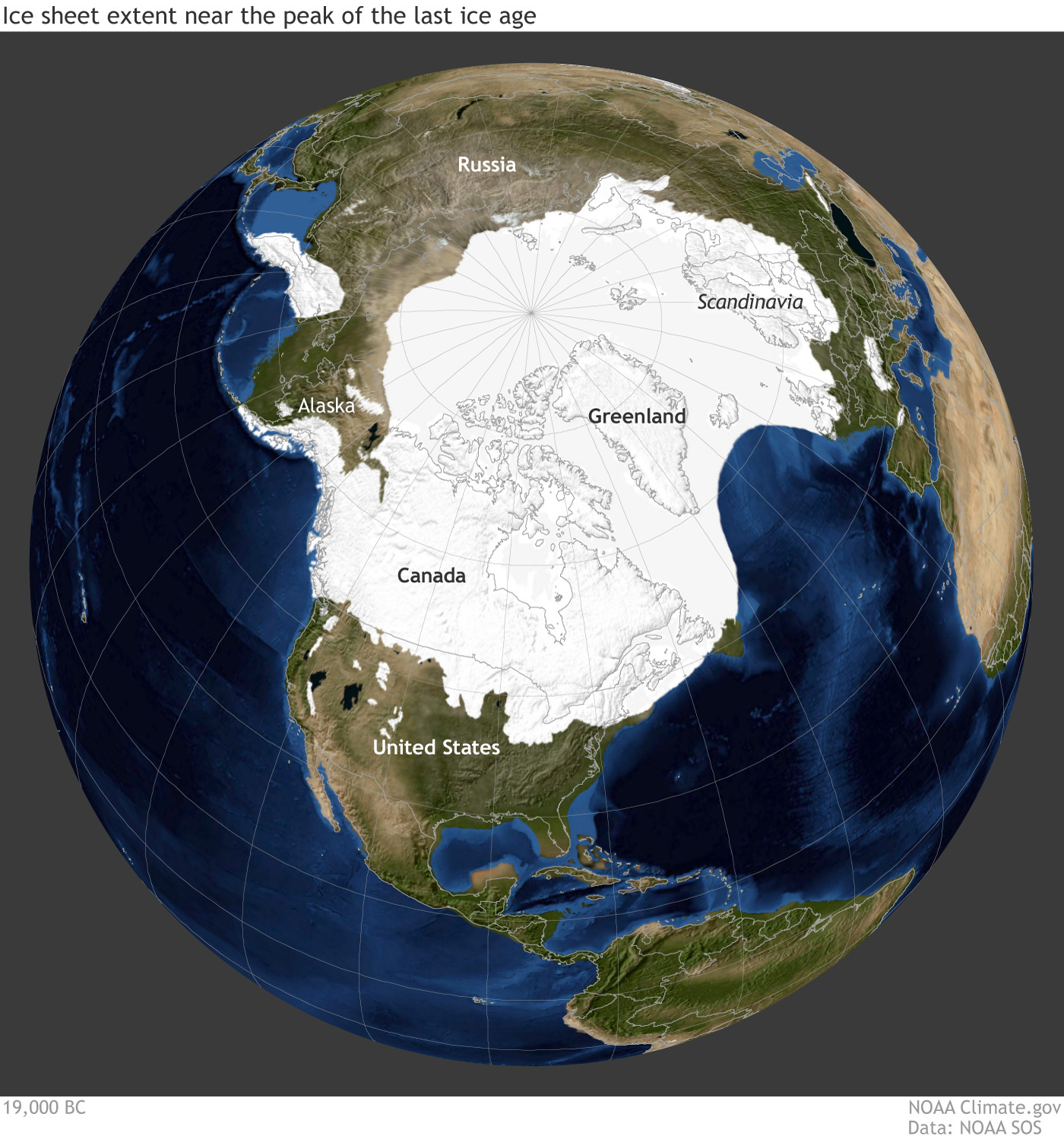 |
By 5000 BCE, the glacier had retreated to more modern levels, leaving ice over the Arctic Ocean and Greenland. CO2 levels were then around 260 ppm. A change of just 100 ppm in CO2 was sufficient to melt the Northern Hemisphere glaciers. The image above is not "Northern Hemisphere-Centric," since the great glaciers were confined to the Northern Hemisphere during the ice ages. That's because glaciers grow over land, and most of the land on the planet is in the Northern Hemisphere. (Our climate studies won't include when one continent - Pangea- existed.) The video below shows an animation of the Northern Hemisphere ice shield from 19,000 BCE to now, with a projected future assuming little action to reduce CO2 emissions. Pay special attention to the graphs, which also show sea level changes.
Best estimates by Tierney et al now show that during the last ice age, the average global temperature was 6 degrees Celsius (11 F) cooler than today, which in the 20th century is 14 C (57 F). The Arctic, however, was much colder (about 14 C or 25 F). The group also came up with an estimate of climate sensitivity, the increase in temperature with increasing CO2. That value is a rise of 3.4 °C (6.1 °F) for a doubling in CO2. In 1896, Arrhenius, recognizing that CO2 was a greenhouse gas, calculated that doubling atmospheric CO2 would cause a rise of 4-5 °C. No one can say we haven't known!
A new value for climate sensitivity, giving a rise of 4.8 °C (8.6 °F) for a doubling of CO2, was determined by Hansen et al (2023) using more precise data about temperatures and CO2 concentrations derived from isotopic analyses of ice core samples. If this value holds, the environment needed for human life and our present society is seriously jeopardized unless we take immediate measures to stop emissions and cool the planet. In addition, the rate of increase in global and ocean temperatures has accelerated after 2010, and the likely culprit is a decrease in toxic SO2 aerosols released by ocean vessels in the North Pacific and Atlantic Oceans, and in China, from the reduction of dirty coal-burning facilities. Aerosols reflect incident solar light and increase cloud formation, both of which help cool the planet. Paradoxically, reducing anthropogenic aerosols, while great for cardiovascular and pulmonary health, will accelerate global warming. India would be much warmer now if not for the high aerosol concentrations from smog that cover many of its large cities and countryside.
Climate, CO2, and temperature have always changed over geological time, but our present rise in anthropogenic CO2 in such a brief time is unprecedented. This has led to CO2 levels far exceeding those during the warmer interglacial periods when Northern Hemisphere glaciers retreated.
The Ice Ages, CO2, and Temperature
It's not CO2 levels that are causing the observed increases in temperature. CO2 levels are rising as temperatures increase, so we don't have to worry about them. It's a natural process requiring no action to reduce fossil fuel use. Why reduce it if it doesn't cause global warming?
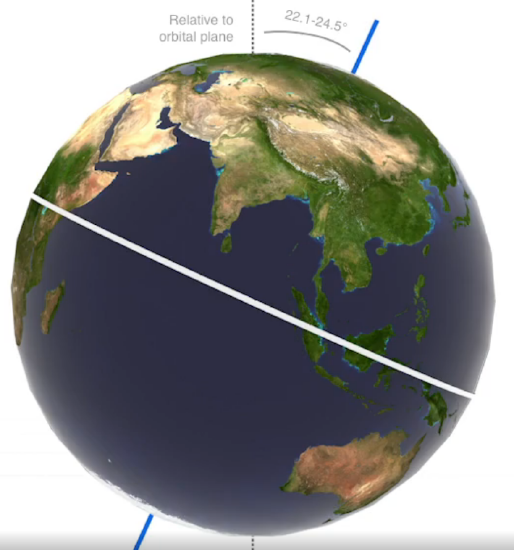
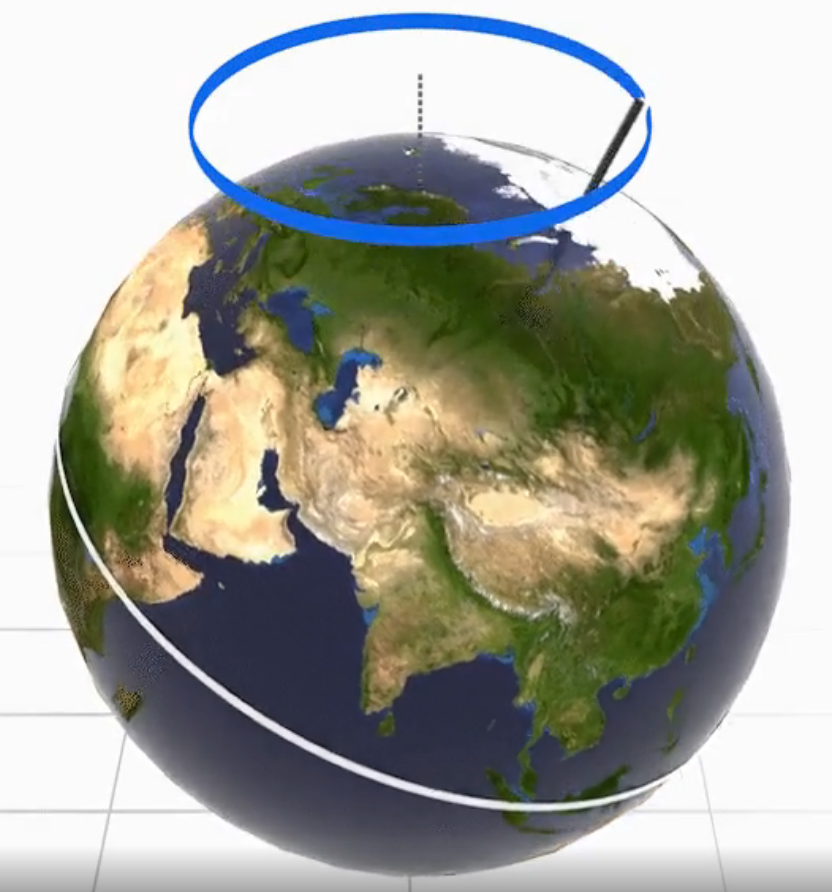
Data and models show that the global increase in temperature is mainly driven by rising CO2 levels (and not rising temperatures driving rising CO2). That begs the question as to what starts the process of deglaciation. It turns out that cyclic increases in solar irradiance that increase temperatures, especially in the Northern Hemisphere, start deglaciation. A prime factor is the changes in the orbital dynamics of the Earth around the Sun. As you know, the orientation of the Earth's rotation axis remains generally fixed and pointed in the same orientation as the Earth rotates around the sun. This fixed orientation leads to Earth's annual spring, summer, fall, and winter cycles. In winter, the Northern Hemisphere is pointed away from the sun, leading to decreased solar irradiance per square meter and causing winter there. When the Earth is on the opposite side of the sun, the axis points in the same direction but tilts towards the sun, leading to summer in the northern hemisphere. However, the Earth's orbital dynamics do change cyclically over long periods. These long-term changes in the Earth's orbital shape (eccentricity), tilt (obliquity), and wobble (precession) are called the Milankovitch Cycle and are illustrated in Figure \(\PageIndex{8}\). These cycles cause small temperature increases that start deglaciation. Click on each image below to download and view short videos illustrating these orbital changes.
Change in eccentricity (orbital shape) - (100,000 yr cycle)
Change in obliquity (tilt) (41,000 yr cycle)
Axle precession (wobble) (26,000 yr cycle)
Figure \(\PageIndex{8}\): The Milankovitch cycle showing changes in the Earth's orbital dynamics with respect to the sun. https://climate.nasa.gov/news/2948/m...arths-climate/
Based on these cycles, Milankovitch calculated that recurring ice ages should occur approximately every 41000 years. Ice ages occurred at this interval, from about 3 million years ago (MYA) to 1 million years ago (MYA). About 800,000 years ago (mid-Pleistocene), they lengthened to about 100,000 years, corresponding to the Earth's eccentricity cycle. The increased cycle duration led to longer-lasting glaciers that moved further south into the Northern Hemisphere. This shift can't be accounted for solely by Earth's orbital eccentricity, since the change in solar irradiance wouldn't have been sufficient to cause it.
CORRECTION - 3/23/26: One likely explanation for the increase in time between ice ages during this Mid-Pleistocene Transition (MPT) was proposed by John A. Clark et al. It goes like this. Before extensive glaciation occurred, the outer surface of the earth was exposed to significant weathering. This converted the outer rock layer in the Northern Hemisphere into broken rock, dirt, and dust, collectively called regolith. When ice sheets initially formed on this, the ice more readily slid over it (lower frictional resistance), leading to faster ice-shield spread and thinner ice shields that more easily melted during deglaciation. The formation/melting of the ice shields was mostly affected (forced) by the obliquity of Earth's axis, which changed over a 40,000-year cycle.
Over time, the soft regolith was eroded and transported by the glaciers as they extended. This left bare rock. Subsequent cycles of glaciation formed ice shields that moved more slowly and thus became thicker. These required longer melting times, supporting a shift to a 100 Kyr cycle. The glaciers then grew over 2 or more obliquity cycles. Evidence supporting this includes the absence of ancient regolith and the exposure of bedrock in Canada and Scandinavia. Punctuating these rhythmic orbital and ice-age cycles are other events, such as large volcanic eruptions and asteroid impacts, that can produce minor to major climate changes and resulting mass extinctions.
![]() March, 2026. New ice core data from Antarctica offers another explanation for the 40Kyr-to-100Kyr transition during the Mid-Pleistocene and supports the role of the oceans in storing and releasing heat. These findings increase our understanding of our present climate change as well. Marks-Peterson et al. analyzed ice-core data from the Allan Hills Blue Ice Area (BIA) that formed over a continuous 3-million-year period. (These types of analyses will be discussed in subsequent chapter sections.) They could directly measure CO2 and methane in individual ice core layers and determine the age of each vertical layer using ⁴⁰Ar dating. An old, long ice core is difficult to analyze because ice pressure fuses layers, making measurements more challenging. Given this compression, in this analysis, an entire glaciation cycle would be compressed into a single layer.
March, 2026. New ice core data from Antarctica offers another explanation for the 40Kyr-to-100Kyr transition during the Mid-Pleistocene and supports the role of the oceans in storing and releasing heat. These findings increase our understanding of our present climate change as well. Marks-Peterson et al. analyzed ice-core data from the Allan Hills Blue Ice Area (BIA) that formed over a continuous 3-million-year period. (These types of analyses will be discussed in subsequent chapter sections.) They could directly measure CO2 and methane in individual ice core layers and determine the age of each vertical layer using ⁴⁰Ar dating. An old, long ice core is difficult to analyze because ice pressure fuses layers, making measurements more challenging. Given this compression, in this analysis, an entire glaciation cycle would be compressed into a single layer.
The data covered a different time sampling than the 800,000-year continuous ice core record (from EPICA Dome C and others) shown in Figure 4 above, which shows CO2 changing from about 180 ppm during glacial maxima to ~280 ppm during interglacial periods, a net change of ~80-100 ppm. The paper examined whether the long-term baseline (a center with periodic increases and decreases) for CO2 and CH4 changed over 3 million years, but with less time resolution due to compression as described above.
The analysis showed no significant variation in average CO2 across the Mid-Pleistocene transition, as CO2 remained consistently low. The same finding was obtained for the Pliocene-Pleistocene Transition (~2.6 MYa) when CO2 was about 250 ppm. CO2 declined by only about 20 ppm (from roughly 250 ppm to 230 ppm) across the entire Pleistocene. Methane did not change. Yet average temperatures dropped more than 2 0C globally and 3.5 0C in the Antarctic. So, relative stability in CO2 occurred from the start of Northern Hemisphere glaciation (around 2.6 MYA at the start of the Pleistocene) and the Mid-Pleistocene transition. Hence, CO2 changes during these times can't solely contribute to changes in glaciation cycles from 40KYr to 100KYr..
During these times, the extent of the Northern Hemisphere ice sheets increased dramatically. Hence, other factors contribute to cooling during these transitions. (Reminder: the Pleistocene Epoch,2.6 million–11,700 years ago, is the period of the ice ages, while the Holocene (11,700 years ago–present) is the current warm period, although many think we are in a new Epoch called the Anthropocene, representing human-driven environmental changes.) The pre-Industrial Revolution average CO2 level of around 280 falls within the Pleistocene average range determined in the paper. Our present value of over 420 is disastrously high and without precedent over 3 million.
The authors suggest that over time frames of millions of years (not the recurrent glaciation cycles over the last 800 MYr, which were driven by orbital and regolith changes), other factors, such as changes in the ocean circulation, increases in ice sheet reflection (albedo), feedback caused by plants, and additional geological changes. CO₂ remains tightly coupled to temperature on orbital timescales (the glacial cycles), but the long-term cooling trend appears to have been driven by other mechanisms. The data is consistent with the regolith explanation offered above for the 40KYr to 100KYr change in glaciation cycles.
Figure \(\PageIndex{9}\) shows how a combination of tilt angle, precession axis, and orbital shape at around 200 KYA (narrow rectangle across all the graphs) combined to lead to low glacial ice volume (bottom graph).
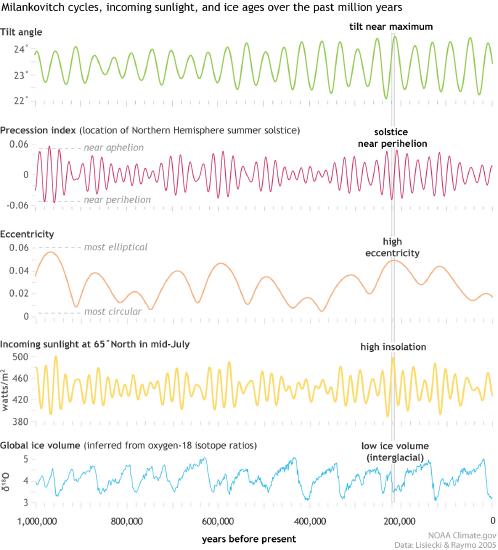
Figure \(\PageIndex{9}\): Milankovitch cycle contribution to ice volume over the past 1M years
If orbital changes (or forcing) trigger deglaciation, what is the role of increasing levels of the greenhouse gas CO2, which covary with temperature (see Figure 3)? Temperature increases derived from orbital and hence solar "forcing" seem to precede CO2 increases for short periods (perhaps 100 - 200 years). After that, CO2 accounts for almost all of the global temperature increase during deglaciation, with CO2 and temperature rising together. A global increase of about 0.3 °C due to the Milankovitch cycle leads to greater Northern Hemisphere irradiance. This causes some localized melting of the Northern ice shield, leading to higher ocean temperatures in the Northern Hemisphere. These increases slow a major ocean current, the Atlantic Meridional Overturning Circulation (AMOC), inhibiting the burial and return of cold water in tropical and southern oceans. This, in turn, led to a warming in the south, accompanied by the release of large amounts of CO2 stored in the oceans (see Carbon Cycle in 31.3). The release of this greenhouse gas was then responsible for most of the warming that led to massive degradation. This "interhemispheric see-saw" heat transfer from northern to southern waters is key. For most of the warming during glacial melting, CO2 and temperature change synchronously.
Recent Updates: April 16, 2026
The graphic information and explanation of the AMOC in Figure \(\PageIndex{10}\) below were made using Claude AI.
Figures \(\PageIndex{10}\)The AMOC. Anthropic. (2025). Claude (Claude Sonnet 4.6) [Large language model]. Retrieved May 2, 2025, from https://claude.ai.
![]() April 15, 2026
April 15, 2026
Unfortunately, a recent report shows the AMOC has a 50% chance of weakening over this century. Here is the abstract and reference to the paper:
"Climate models show considerable discrepancies in their future projections around the Atlantic, mainly due to uncertainties in the fate of the Atlantic Meridional Overturning Circulation (AMOC). Climate models suggest a 32 ± 37% reduction in AMOC strength by 2100 (90% probability, Shared Socioeconomic Pathways 2-4.5 scenario). [The newer, most refined model] gives an estimate of the AMOC slowdown of 51 ± 8% (90% probability), i.e., a weakening ∼ 60% stronger than suggested by the multimodel mean. This more substantial AMOC weakening has key implications for future adaptation strategies. Valentin Portmann et al., Observational constraints project a ~50% weakening of the AMOC by the end of this century. Sci. Adv. 12, eadx4298 (2026).DOI:10.1126/sciadv.adx4298
Here are some consequences of a slowing of the AMOC:
- a cooler, stormier climate, drier summers, and wetter winters in Europe, counteracting some of the warming in that area;
- the accumlation of warm water on the eastern North American coast, causing higher sea level rise compared to other regions (as warm water has a greater volume);
- major disruption of tropical rains, causing flooding and droughts in different regions, including the Amazon and tropical Africa;
- a strong decline in ecosystem mass over the North Atlantic, affecting the size and location of fish;
- a dangerous positive feedback loop of less CO2 deposited and sequestered in the ocean, increasing atmospheric CO2;
- changes in the jet stream with stronger westerly winds in mid-latitudes and an extension of the jet stream over the North Atlantic, having significant effects on weather and climate;
- In a more positive effect, the Arctic ice cap would likely expand.
Interpreting climate data is difficult. For example, it was found, by measuring 15N/14N ratios, that gases like N2 and, by extension, CO2 could rapidly diffuse through the compacting snow (firn, comprising the top 50-100 meters of the ice cap) until they became trapped in the solid ice beneath it. This would lead to "newer" CO2 in older ice samples, and the conclusion that temperature changes preceded changes in CO2. The data are corrected to address the "apparent" time shift.
The CO2 trapped in bubbles in Antarctic ice core samples reflects global CO2 levels, given atmospheric circulation. However, the temperatures measured from the same core samples (see Chapter 31.2) represent local (Antarctic) temperatures. Ice core samples from Greenland and ocean sediment samples from around the world determine temperatures at different locations over time. All of this data is required to model the climate. Combined, they lead us to our present interpretation of the linkage of CO2 and temperature rise over time.
Increased solar irradiance on Earth, resulting from cyclic changes in the Earth's orbit, leads to short, small temperature increases in the Northern Hemisphere. These increases lead to the release of the greenhouse gas CO2 from the oceans, causing synchronous global warming and subsequent deglaciation.
So when skeptics say that temperature increases precede CO2 increases, you can acknowledge they did, but that the bulk of the warming is attributed to increasing CO2 released from ocean stores, leading to synchronous temperature increases and deglaciation. Using chemical terms, small temperature increases from orbital forcing catalyzed the release of large amounts of dissolved CO2 from the ocean. Chapter 31.3 will explore the carbon cycle in more detail and examine how it affects CO2 levels.
Recent Updates: April 10, 2026
This is the argument of a preeminent physicist with expertise in spectroscopy but not in climate science. He states that the absorption of emitted infrared light by CO2 (atm) has peaked because its concentration is high enough to saturate the absorption (0% transmittance), so extra CO2 would have no effect. If so, we can continue burning fossil fuels without worrying about their effects on global warming. Climate scientists have refuted this idea using three main arguments.
1. Spectral line widening prevents saturation
Beer's Law states that A667 cm-1 or 15 μM = εbc, where c is the concentration. If there is sufficient CO2 to absorb all the IR light at 15 μM, the signal saturates, and additional CO2 won't absorb any more IR light. It would escape into space, with no additional warming of the planet. However, the main absorbance band at 15 μM is wide and becomes wider with increasing CO2, so additional CO2 can still absorb and trap IR at the wavelengths, or "wings," surrounding the main 15 μM peak. This is illustrated in Figures \(\PageIndex{11}\) below.
Figure 11: IR transmission spectra of CO2 with increasing CO2. Drag the sliders to see the band broaden in real time. The dashed blue line shows the pre-industrial baseline (280 ppm) for comparison. Claude (Claude Sonnet 4.6) [Large language model]. Retrieved May 2, 2025, from https://claude.ai.
The main peak becomes broader due to collisions with molecules (most notably O2 and N2). At higher pressure (lower altitude, as in the troposphere), there are more collisions per second, and the wings are wider as the energy levels for the absorption are perturbed. You can see this by dragging the pressure slider — lower pressure (higher altitude) narrows the band. Another factor leading to broadening is the Doppler effect. If a photon and CO2 are moving in different directions during absorption, the photon's frequency changes slightly, leading to band broadening. You likely associate the Doppler effect with a shifting frequency of a siren moving away from you.
It is well known that doubling the concentration of CO2 does not double the temperature. This assumes a linear relationship between the two. Rather, a doubling of CO2 produces a net increase but far less than a doubling of the temperature change, so the relationship is logarithmic in CO2, not linear. This suggests some but not complete saturation of the CO2 infrared absorption. We've explored climate sensitivity, the increase in temperature with increasing CO2, earlier in this section, and showed that a doubling of CO2 gives an increase of 3.4 °C (6.1 °F) or possibly even higher. Climate sensitivity is included in all climate models, so any saturation effect is already accounted for.
2. CO2 is distributed in the upper troposphere and stratosphere
CO2 is not just located close to the ground where it is emitted. In the upper troposphere and stratosphere, its concentration is lower and much less "saturated," and infrared light is still absorbed. Moreover, at these heights, less heat is radiated back into space as colder objects radiate less.
3. Direct satellite data show a decrease in IR radiation from the atmosphere with increasing CO2
This is clearly the "smoking gun" to refute the saturation argument. Figures \(\PageIndex{12}\) below shows the annual mean radiance differences from the Earth due to CO2 increases. The data are from the Atmospheric Infrared Sounder (AIRS) satellite (blue line) and are compared with theory (red line) during 2003–2012.
Figures \(\PageIndex{12}\): Annual mean radiance differences (in mW m−2 sr−1 (cm−1)−1) due to CO2 increase from the AIRS observations (blue line) and from theory (red line) and standard deviations for the AIRS observations (blue shading), illustrating the direct impact of CO2 increase on the spectral radiances during the 2003–2012 period. João Teixeira, R. Chris Wilson, and Heidar Th. Thrastarson. Atmospheric Chemistry and Physics. 24, 6375–6383, 2024. https://doi.org/10.5194/acp-24-6375-2024. Creative Commons Attribution 4.0 License.
Here is the significance of these observations taken directly from the paper: "This figure is focused on the 680-to-780 cm−1 spectral range, which represents the R branch of the 15 µm CO2 band and is a spectral region where the CO2 signal is particularly significant. In this spectral region, enhanced absorption in the troposphere, where temperature decreases with height, reduces outgoing "infrared" radiation (y-axis values are negative). From a broader climate perspective, the reduction in outgoing radiation is behind the increase in global surface temperature that is necessary for the overall climate system to re-establish energy balance at the top of the atmosphere, and as such it is a critical component of global warming. During this period, the measured monthly mean CO2 mole fraction at Mauna Loa increased on average by approximately 2 ppm yr−1".
The main proponent of the saturation argument is leading the present US administration's efforts to refute the overwhelming scientific consensus on the main cause of present climate change: CO2 emitted from burning fossil fuels.
We refute another of his arguments, that increasing CO2 is good for the planet since it is merely plant food, in another section.
Termination of the Ice Ages
How did the ice ages terminate? Contributions from orbital forcing derived from the Milankovitch cycle also play a role. Another factor seems to be dust derived from regolith, produced by glacier movement, as mentioned above. How can that hypothesis be tested? Using proxies for dust, namely iron and long-chain n-alkanes (derived from plant waxes) that have been deposited in sediments. First, look at a graph of CO2 and temperature changes and superimpose those on iron and long-chain fatty acid levels, as shown in Figure \(\PageIndex{13}\).
| |
A close examination of the two vertically aligned graphs from around 120 K to 130 KYA shows that iron and n-alkane depositions are at a minimum while CO2 and temperature are peaking! What explains this negative correlation? It depends on the intimate connection of the biosphere with the nonbiological world (an arbitrary distinction).
Iron and n-alkanes are circulated and delivered in dust. The long-chain alkanes, highly abundant in waxes and enriched in odd-carbon-number chains, were presumably derived from leaf waxes, which prevent water loss from plants, especially at higher temperatures. Dust deposits were first observed in geological time during the transition from the warmer Pliocene (5.3 to 2.6 MYA) to the Pleistocene (2.6 MYA to 11.7 KYA; see Figure 8 below). During the warmer Pliocene, the difference between global and atmospheric temperatures was lower, and with this smaller temperature gradient, winds capable of globally transporting dust would be diminished. Also, the warmer Pliocene (5.3 to 2.6 MYA) would have had more rain, which would have removed dust from the global circulation.
As temperatures cooled in the Pleistocene (2.6 MYA to 11.7KYA), cycles of glaciation would produce more dust-containing regolith (rocks, soil, and dust) at the southern ends of the ice shield, which would be dispersed by stronger global winds driven by higher temperature gradients and less rainfall. Dust contains carbon (for example, long-chain fatty acids) and, perhaps more importantly, iron, which is needed for oceanic phytoplankton growth. Without Fe, the uptake of CO2 by phytoplankton (primary production) would not occur, leading to increased CO2 in the atmosphere. Stronger regional atmospheric winds would lead to increased nutrient upwelling and deep-ocean CO2 release. The CO2 would enter the atmosphere more readily in the absence of dust deposition of iron.
In summary:
- High CO2 and high temperature (lower global temperature gradients, more rain) are associated with lower dust levels, as indicated by proxies Fe and n-alkanes. Low dust levels lead to lower deposition of Fe and n-alkanes in the ocean, which decreases phytoplankton primary production and the fixation of CO2 into biomass. This leads to increased CO2 movement from the ocean to the atmosphere, increasing temperature. This is an example of a positive feedback loop (higher temperatures leading to higher temperatures).
- Low CO2 and low temperature (higher global temperature gradients, stronger winds, less rain) are associated with high dust deposition of Fe and n-alkanes. This increases phytoplankton primary production and decreases CO2 flux from the ocean to the atmosphere, forming a negative feedback loop.
By the end of a glacial deposition cycle, dust, blown by stronger winds from higher temperature gradients, was increasingly deposited on the ice sheets. In addition to increasing heat absorption by the sheets, it would also decrease their reflectivity (albedo). Both effects would promote ice sheet melting. Also, a cooler planet during the glacial maximum had less precipitation, which, along with lower CO2, would lead to more plant and tree death, increasing soil erosion and desertification, both effects which would have increased dust production and its deposition on ice sheets. Then, when CO2 rose to 280 ppm, plant life renewed, and dust levels dropped.
Climate change from 66 million years ago to now
Antarctic ice core data cover the last 2 million (M) years. Ocean sediment data can go back 66 million years (MYA), just before the dinosaurs died following the massive asteroid impact that formed the Chicxulub crater, buried beneath the Yucatán Peninsula in Mexico. A brief review of geological eras, periods, and epochs is shown below in Figure \(\PageIndex{14}\)
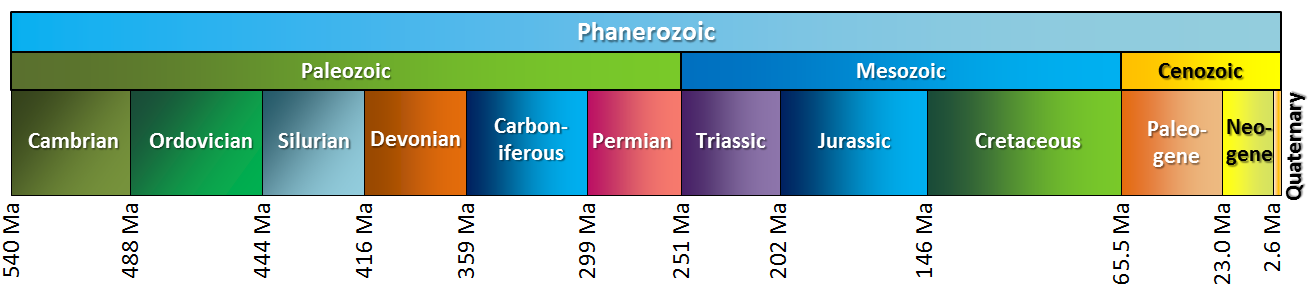
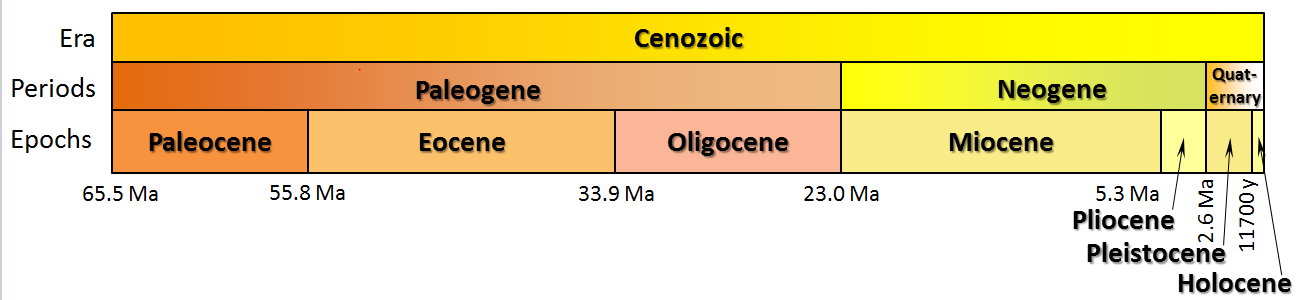
Figure \(\PageIndex{14}\): Geological Era, Periods and Epochs
CO2 levels and associated temperatures derived from ocean sediment cores going back to 66 MYA are shown in Figure \(\PageIndex{10}\).
Figure \(\PageIndex{14}\) CO2 levels (red) and temperatures (blue) derived from ocean sediment cores going back to 66 MYA = 66,000 KYA . Data from Rae et al. Annual Review of Earth and Planetary Sciences, 49, 2021
Again, note the parallel rise and fall of CO2 and temperature. Eventually, they fell further in the Pliocene (5.3 to 2.6 MYA) and Pleistocene (2.6 MYA to 11.7KYA) epochs with cyclic glacier/interglacial periods we've discussed above. Northern hemisphere glaciation started in the late Miocene (10 to 6 MYA), and both poles of the planet had glacial sheets.
The time frame shown in Figure 9 encompasses the Cenozoic era (65 MYA when the dinosaurs died to about now). CO2 levels were much higher than today in the greenhouse Paleocene and Eocene eras, but decreased to about 500 ppm in the Oligocene (34 MYA). An almost stepwise drop in CO2 and temperature occurred in the Eocene to Oligocene transition (EOT), about 33 MYA. Data shows the development of large ice sheets in Antarctica at that time. Before the EOT (33 MYA), Antarctica was ice-free, as shown in the recreation in Figure \(\PageIndex{15}\).

Figure \(\PageIndex{15}\): Reconstruction of the West Antarctic mid-Cretaceous temperate rainforest. Image credit: J. McKay / Alfred-Wegener-Institut / CC-BY 4.0. https://www.sci.news/othersciences/p...ing-09921.html
Proxy temperature data indicate that the transition was most likely driven by decreased CO2, with some orbital forcing probably involved. Present models still struggle to explain the EOT (33 MYA) transition, but it is clear that both CO2 and temperature decreased. Where did the CO2 go? Most assuredly into the oceans.
To understand that, we have to understand a bit about the carbon cycle, which we will discuss more fully in the next chapter section. Let's briefly discuss the role of atmospheric CO2 and its interaction with the ocean. The main atmospheric gases, N2 and O2, are present at very low concentrations in the ocean because they are nonpolar and generally unreactive. CO2 is also a nonpolar trace gas, but unlike other nonpolar gases, it can readily react with water to form HCO3- and CO3-2, which are abundant in ocean reserves. Hence, ocean chemistry of CO2 largely determines atmospheric CO2 levels. The coupled reactions of CO2 are shown below.
\begin{equation}
\mathrm{CO}_2(\mathrm{~g}, \mathrm{~atm}) \leftrightarrow \mathrm{CO}_2(\mathrm{aq}, \text { ocean) }
\end{equation}
\begin{equation}
\mathrm{CO}_2(\mathrm{aq} \text {, ocean })+\mathrm{H}_2 \mathrm{O}(\mathrm{I} \text {, ocean }) \leftrightarrow \mathrm{H}_3 \mathrm{O}^{+}(\mathrm{aq})+\mathrm{HCO}_3^{-}(\mathrm{aq})
\end{equation}
\begin{equation}
\mathrm{H}_2 \mathrm{O}(\mathrm{I})+\mathrm{HCO}_3^{-}(\mathrm{aq}) \leftrightarrow \mathrm{H}_3 \mathrm{O}^{+}(\mathrm{aq})+\mathrm{CO}_3{ }^{2-}(\mathrm{aq} \text {, sparingly soluble })
\end{equation}
This chemistry helps determine the ocean's pH. Figure \(\PageIndex{16}\) shows atmospheric CO2 and ocean pH levels over the last 66 million years.
V3GraphCO2_pH.png?revision=1&size=bestfit&width=963&height=577)
Figure \(\PageIndex{16}\): Atmospheric levels of CO2 and ocean pH over the last 66 million years
Before the EOT at 34 MYA, atmospheric CO2 levels were higher and ocean pH levels lower (around 7.7). After the EOT (33 MYA), atmospheric CO2 is much lower, and ocean pH is higher (more basic, 7.9, rising to 8.1). What happened to the CO2 is a bit unclear. Atmospheric CO2 decreased as it moved into the oceans, but wouldn't that have lowered pH based on the chemical equations presented above? It would have, but it turns out that the ocean alkalinity is determined not just by H3O+ produced by the equations above but by the dissolved inorganic carbon ions, HCO3- (aq) and CO32- (aq), which are conjugate bases. Increased HCO3- (aq) and SiO4-2 (aq) from weathering solid carbonates and silicates that entered the oceans would raise ocean pH.
A little review of introductory chemistry helps here.
Let's take bicarbonate, the weak conjugate base of the weak acid carbonic acid. HCO3- can act as both an acid and base.
Rx 1: Acts as an acid: HCO3- (aq) + H2O (l) ↔ H3O+(aq) + CO32- (aq)
\begin{equation}
K_{a 2}=\frac{\left[\mathrm{H}_3 \mathrm{O}^{+}\right]\left[\mathrm{CO}_3^{2-}\right]}{\left[\mathrm{HCO}_3^{-}\right]}=4.7 \times 10^{-11}
\end{equation}
Rx 2: Acts as a base: HCO3- (aq) + H2O (l) ↔ H2CO3 (aq) + OH- (aq)
\begin{equation}
K_{b 2}=\frac{\left[\mathrm{H}_2 \mathrm{CO}_3\right]\left[\mathrm{OH}^{-}\right]}{\left[\mathrm{HCO}_3^{-}\right]}=2.2 \times 10^{-8}
\end{equation}
The equilibrium constant for the reaction of HCO3- as a base is much larger, so bicarbonate is a stronger base than an acid.
Whatever the mechanism of CO2 drawdown, it led to lower temperatures during the EOT transition. The ocean's increased alkalinity also consumed H3O+, increasing ocean pH.
Figure \(\PageIndex{17}\) summarizes planetary temperatures across geological time.
Figure \(\PageIndex{17}\): Earth's temperature over 500 million years. https://commons.wikimedia.org/wiki/F...alaeotemps.png. (Excel available). Creative Commons Attribution-Share Alike 3.0 Unported
Several key features should be noted. The last time CO2 was as high as today (415 ppm) was about 3 million years ago. Repetitive cycles of glaciation/deglaciation are obvious in the Pleistocene (2.6 MYA to 11.7 KYA). Note: This figure has been misinterpreted, leading to misinformation in the popular press and some podcasts, as it shows a general decrease in atmospheric CO2 levels since the end of the dinosaurs (65 MYA). It shows that our current CO2 levels most closely resemble those of 3-4 million years ago. At that time, Earth's temperature was likely 3 °C above what it is today, and the seas were 25 meters higher than they are today. In addition, the steep slope of the present rise is unprecedented in the year's historical record.
At around 56 MYA, a temperature spike of about 50 - 9 0F (or even more) on an already warmer planet occurred over about a 100 K-year timeframe. This is known as the Paleocene/Eocene thermal maximum (PETM). It is the closest example to the current rate of CO2 injection into the atmosphere, so it gives us a basis for what we may see with our current CO2 spike. The poles warmed to nearly 70 0F, and alligators and palm trees were found there. The warming also led to the spread of tropical rainforests from the equator, allowing the evolution and proliferation of new plant species, including flowering plants and an incredible biodiversity of insects, birds, and animals that relied on them. Flowering plants produce fruits, which helped drive the evolution of mammals and the first true primates, including the tiny Teilhardina magnoliana. It may have resembled the picture in Figures \(\PageIndex{18}\) below. (https://en.wikipedia.org/wiki/Teilhardina). Big eyes and hands would help them find fruit in tropical forests.
Figures \(\PageIndex{18}\): Teilhardina magnoliana
The temperature increase was caused by a dramatic spike in CO2 from deep-sea volcanoes and vents, which also led to a dramatic drop in ocean pH as measured by the loss of deep-sea CaCO3 (chalk). Methane hydrates were also released. These environmental and biosphere changes are visually evident in geological deep-sea sediment records as shown in Figure \(\PageIndex{19}\).
Figure \(\PageIndex{19}\): Overview of the Paleocene–Eocene Thermal Maximum (PETM, 55.5 MYA) data from deep-sea records and the terrestrial Polecat Bench
(PCB) drill core against age. Westerhold et al. Clim. Past, 14, 303–319, 2018. https://doi.org/10.5194/cp-14-303-2018. Creative Commons Attribution 3.0 License
Sediment cores were taken at various sites (1262, 1267, 1266, 1265, 1263, and 690), aligned from left to right according to water depth, from deep to shallow. Note that 55.93 million years ago, at the start of the PETM, there was a sharp transition from light brown/gray, enriched in chalk, to dark brown, enriched in clay. Ocean acidification dissolved the chalk. It took over 100,000 years to recover.
This very short time frame is called the Paleocene/Eocene thermal maximum (PETM,55.5 MYA), which shows very quick spikes (on the geological time scale) can and do occur. Approximately 1.5 petagrams (1015) of CO2 were released annually during the PETM. We are now releasing about 25 petagrams per year. Our current rate of warming is much higher than during the PETM (55.5 MYA). The best candidates for the source of CO2 responsible for the PETM are volcanoes, the oceans, and permafrost. Methane hydrates (a solid form of methane found in low-temperature, high-pressure waters) might also be another factor.
The PETM and time after allowed for the optimal evolution of primates. That time might be called the Age of the Primates (analogous to the Age of the Dinosaurs). Figure 13 shows that the arrival of the Oligocene was accompanied by much lower temperatures resulting from CO2 drawdown. This decimated primate habitats, except in tropical rainforests. Primates disappeared completely from North America but thrived in Africa. Tectonic forces generated the African Rift Valley, characterized by diverse geology and a less homogeneous, more fractured landscape, which again posed new challenges but also provided new habitats for the evolution of primates and the eventual appearance of Homo Sapiens.
Update from December 3, 2023, Science
In December 2023, a refinement of CO2, temperature, and sea-level-rise data since the demise of the dinosaurs 65 million years ago was published. (The geological interval from 66 million years ago to now is called the Cenozoic Era and consists of 3 periods: the Paleogene until 23 MYA, the Neogene until 2.6 MYA, and the Quaternary.) A group, the Cenozoic Carbon Dioxide Proxy Integration Project (CenCO2PIP) Consortium, reviewed and reevaluated all "proxy" measurements used to determine past climate conditions (we will discuss proxies using isotope ratios in ice cores and sediments as proxies for actual CO2, temperature, and sea level changes in the next few sections). Figure \(\PageIndex{20}\) below shows the revised data, representing our best. For this work, pay close attention to graphs A (CO2), B (relative temperature changes), and C (sea level changes). Note that CO2 levels in 2022 were last seen 14 million years ago!
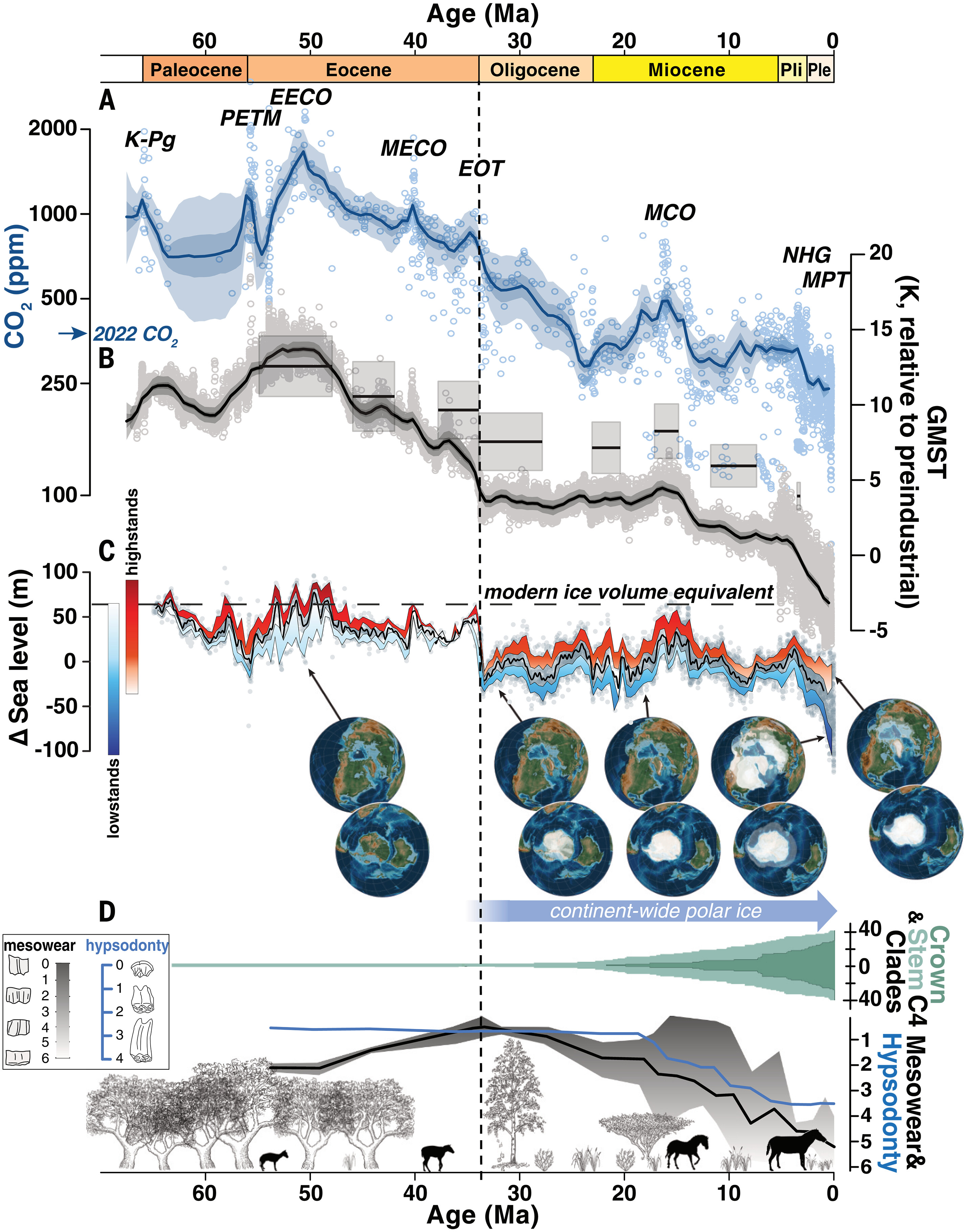
Figure \(\PageIndex{20}\): Category 1 paleo-CO2 record compared to global climate signals. From The Cenozoic CO2 Proxy Integration Project (CenCO2PIP) Consortium, Toward a Cenozoic history of atmospheric CO2.Science382, eadi5177(2023). DOI:10.1126/science.adi5177. Reprinted with permission from AAAS.
The legend below is directly taken from the Science article. The vertical dashed line indicates the onset of continent-wide glaciation in Antarctica.
(A) Atmospheric CO2 estimates (symbols) and 500-kyr mean statistical reconstructions (median and 50 and 95% credible intervals: dark and light-blue shading, respectively). Major climate events are highlighted: K-PG, Cretaceous/Paleogene boundary; PETM, Paleocene Eocene Thermal Maximum; EECO, Early Eocene Climatic Optimum; MECO, Middle Eocene Climatic Optimum; EOT, Eocene/Oligocene Transition; MCO, Miocene Climatic Optimum; NHG, onset of Northern Hemisphere Glaciation; and MPT, Mid-Pleistocene Transition. The 2022 annual average atmospheric CO2 of 419 ppm is indicated for reference.
(B) Global mean surface temperatures estimated from benthic δ18O data following Westerhold et al. (solid line, individual proxy estimates as symbols, and statistically reconstructed 500-kyr mean values shown as the continuous curve, with 50 and 95% credible intervals) and from surface temperature proxies (gray boxes).
(C) Sea level after with gray dots displaying raw data; the solid black line reflects median sea level in a 1-Myr running window. High- and lowstands are defined within a running 400-kyr window, with lower and upper bounds of highstands defined by the 75th and 95th percentiles and lower and upper bounds of lowstands defined by the 5th and 25th percentiles in each window. Globes depict select paleogeographic reconstructions and the growing presence of ice sheets in polar latitudes.
(D) Crown ages show that C4 clades, with CCMs adapted to low CO2, initially diversified in the early Miocene, and then rapidly radiated in the late Miocene. Flora transition from dominantly forested and woodland to open grassland habitats based on fossil phytolith abundance data. North American equids typify hoofed animal adaptations to new diet and environment, including increasing tooth mesowear (black line; note the inverted scale), hypsodonty (blue line), and body size.
The results reconfirm the strong correlation across geologic time of CO2 and temperature, but there are some time intervals when they were out of synchrony. One example is from 37 to 34 MYA, around the Eocene to Oligocene transition (EOT), described above. In Figure 15, CO2 didn't change much as the Earth cooled prior to the EOT. In contrast, CO2 levels dropped sharply in the Oligocene as temperatures remained flat. The start of the Oligocene also saw a large drop (55 m) in sea levels, coincident with the EOT, the fall in temperatures, and the rise in glaciation. The authors state that some of the divergences in CO2 and temperatures are likely not directly related to CO2 but to other variables, such as changes in "paleogeography" arising from continental drift, plate tectonics, etc., that might have altered ocean currents, as well as changes in the reflectivity of sunlight. In addition, the proxies used for those times might have been affected by unaccounted-for changes in seawater composition and pH.
This extensive data presented an opportunity of calculate the climate sensitivity (how much temperature changes with a doubling of CO2) as we discussed above. The value calculated by Tierney was about 3.4 °C (6.1 °F), although Hansen's recent estimate is higher. This particular value is the equilibrium climate sensitivity (ECS), which includes relatively fast feedbacks like cloud cover and sea ice that act over 100s of years. The value calculated in the new study is not as fine-grained (coarser) at resolutions of 1000s of years, so instead they calculate a much longer-term climate sensitivity called the Earth Systems Sensitivity (ESS) that includes long-term (geological time) events such as changes in continent-wide ice sheets. The value of ESS is between 5°C (9°F) to 8°C (14.4°F), much higher than the ECS widely accepted for our present time. A doubling of CO2 without some mechanism for drawdown, if sustained over geological time, would transform the world of the distant future. It would be similar to conditions during the Paleocene/Eocene thermal maximum (PETM), when the poles warmed to near 70 0F, allowing alligators and palm trees to occur there.
This chapter is divided into two sections. Read Chapter 01B, "Back to the Present and Future of Climate Change," to see where we are and where we are headed!
Summary
This chapter introduces the fundamental science behind climate change, focusing on how human activities have altered the Earth’s energy balance and disrupted long-established climate patterns. Key points include:
-
Historical Foundations and Urgency:
- Early studies by Eunice Foote and John Tyndall laid the groundwork for understanding the greenhouse effect.
- Modern climate models (developed since the 1980s) and recent observations confirm that anthropogenic CO₂ emissions are driving rapid global warming, necessitating immediate and aggressive action.
-
The Greenhouse Effect and Energy Imbalance:
- Earth’s climate is maintained by a balance between incoming solar radiation and outgoing infrared radiation.
- Greenhouse gases such as CO₂, methane (CH₄), and nitrous oxide (N₂O) absorb infrared radiation, effectively insulating the planet.
- Pre-industrial CO₂ levels (~280 ppm) have risen to 427 ppm as of January 2025, upsetting this balance and storing excess energy primarily in the oceans.
- The current energy imbalance is significant—equivalent to detonating several Hiroshima-type atomic bombs every second—illustrating the unsustainability of current trends.
-
Global Warming Potential (GWP):
- GWP is a metric used to compare the warming effects of different greenhouse gases in CO₂-equivalents over a 100-year period.
- Although CH₄ is more potent on a per-mass basis over short time frames, CO₂ remains the principal concern due to its long atmospheric lifetime and cumulative impact.
-
Climate Variability Over Geological Time:
- Ice core data reveal that while CO₂ and temperature have fluctuated naturally over the past 800,000 years, the recent rapid rise in CO₂ levels is unprecedented.
- The chapter discusses glacial-interglacial cycles, highlighting how relatively small changes in CO₂ (100 ppm) historically corresponded with major shifts in global climate.
-
Orbital Forcing and Feedback Mechanisms:
- The Milankovitch cycles (changes in Earth’s eccentricity, obliquity, and precession) initiate small temperature increases that trigger deglaciation.
- These temperature rises lead to the release of CO₂ from the oceans, amplifying warming through positive feedback loops.
- Dust feedbacks are also explored, showing how variations in dust deposition can either enhance or mitigate warming by influencing phytoplankton growth and planetary albedo.
-
Long-Term Climate Change and Carbon Cycle Interactions:
- The chapter reviews data spanning from 66 million years ago to the present, illustrating how CO₂ levels and global temperatures have paralleled each other over geological time.
- Major events such as the Paleocene–Eocene Thermal Maximum (PETM) are discussed, providing context for the rapid changes occurring today.
- The interactions between atmospheric CO₂ and ocean chemistry are detailed through chemical equilibria, explaining shifts in ocean pH and alkalinity that accompany changes in CO₂ concentrations.
-
Implications for Future Climate:
- Updated data indicate that equilibrium climate sensitivity (ECS) and the longer-term Earth Systems Sensitivity (ESS) may be higher than previously estimated, suggesting that a doubling of CO₂ could eventually raise global temperatures by 5–8 °C.
- The chapter concludes by emphasizing the critical need to address CO₂ emissions to avoid future climate scenarios that could jeopardize human habitability and ecosystem stability.
Overall, this chapter provides a comprehensive framework that combines historical context, physical principles, and biochemical processes to explain current climate change. It lays the groundwork for understanding how climate dynamics are interconnected with global biogeochemical cycles, setting the stage for further exploration of climate mitigation and adaptation strategies in subsequent sections.