3.1: Internal Energy
- Page ID
- 9540
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)3.1.1. Definitions
In thermodynamics, internal energy consists of the sum of microscopic (molecular scale) kinetic and potential energy.
Microscopic kinetic energies include random movement (translation) of molecules, molecular vibration and rotation, electron motion and spin, and nuclear spin. The sum of these kinetic energies is called sensible energy, which we humans can sense (i.e., feel) and measure as temperature.
Microscopic potential energy is associated with forces that bind masses together. It takes energy to pull two masses apart and break their bonds. This is analogous to increasing the microscopic potential energy of the system. When the two masses snap back together, their microscopic potential energy is released back into other energy forms. Three forms of binding energy are:
- latent — bonds between molecules
- chemical — bonds between atoms
- nuclear — sub-atomic bonds
We will ignore chemical reactions and nuclear explosions here.
Latent energy is associated with phase change (solid, liquid, gas). In solids, the molecules are bound closely together in a somewhat rigid lattice. In liquids, molecules can more easily move relative to each other, but are still held close together. In gases, the molecules are further apart and have much weaker bonds.
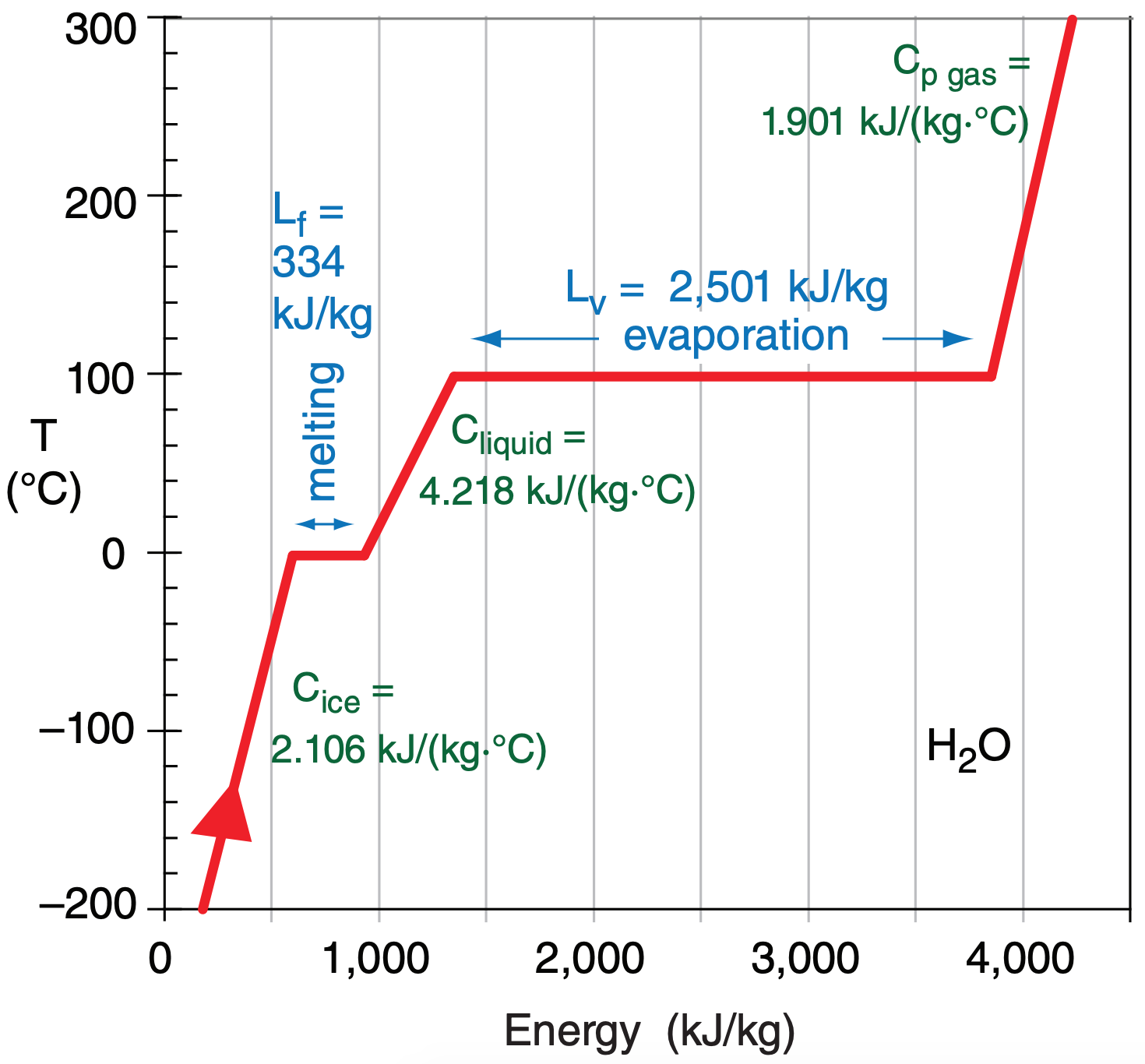
For example, starting with cold ice (in lower left corner of Figure 3.1), adding energy causes the temperature to increase (a sensible effect), but only up to the melting point (0°C at standard sea-level pressure). Further addition of energy forces bonds of the solid lattice to stretch, enabling more fluid movement of the molecules. This is melting, a latent effect that occurs with no temperature change. After all the ice has melted, if you add more energy then the liquid warms (a sensible effect), but only up to the boiling point (100°C). Subsequent addition of energy forces further stretching of the molecular bonds to allow freer movement of the molecules; namely, evaporation (a latent effect) with no temperature change. After all the liquid has vaporized, any more energy added increases the water-vapor temperature (a sensible effect).
Figure 3.1 can also be traversed in the opposite direction by removing internal energy. Starting with hot water vapor, the sequence is cooling of the vapor, condensation, cooling of the liquid, freezing, and finally cooling of the ice.
3.1.2. Possession and Transfer of Energy
We cannot ignore the connection between the microscale and the macroscale. A macroscale object such as a cannon ball consists of billions of microscale molecules and atoms, each possessing internal energy. Summing over the mass of all the molecules in the cannon ball gives us the total internal energy (sensible + latent energy) that the cannon ball possesses.
Thermal energy transferred to or from the macroscale object can increase or decrease the internal energy it possesses. This is analogous to your bank account, where money transferred (deposited or withdrawn) causes an increase or decrease to the total funds you possess.
3.1.2.1. Transfer of Heat
Define the transfer of thermal energy as ∆q. It has energy units (J kg–1). In this text, we will refer to ∆q as heat transferred, although in engineering texts it is just called heat.
3.1.2.2. Latent Energy Possessed
Define the latent heat QE as the latent energy (J) possessed by the total mass m of all the molecules in an object.
But usually we are more interested in the change of possessed latent heat ∆QE associated with some process that changes the phase of ∆m kilograms of material, such as phase change of water:
\(\ \begin{align}\Delta Q_{E}=L \cdot \Delta m_{\text {water}}\tag{3.1}\end{align}\)
For example, if we transfer ∆q amount of thermal energy into water that is already at 100°C at sea-level pressure, then we can anticipate that the amount of water evaporated will be given by: ∆mwater = ∆q·mwater/Lv. [A sample application is on page 86.]
Different materials have different strengths of bonds, so they have different constants of proportionality L (called the latent heat factor) between ∆QE and ∆m. For water (H2O), those latent heat factors are given in Table 3-1.
3.1.2.3. Sensible Energy Possessed
The sensible energy (J) possessed by the total mass m of all the molecules in an object (such as air molecules contained in a finite volume) is mair·Cv·T, where T is absolute temperature of the air. The constant of proportionality Cv is called the specific heat at constant volume. Its value depends on the material. For dry air, Cvd air = 717 J kg–1 K–1.
Again, we are interested more in the change of sensible energy possessed. We might suspect that it should be proportional to the change in temperature ∆T, but there is a complication that is best approached using the First Law of Thermodynamics.


