7.1.3: Ease of Weathering
- Page ID
- 18688
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Some minerals break down more easily than others. Geologists can compare weathering rates by looking at minerals in rock outcrops, and by studying the minerals present in sediments of different ages. Goldich (1938) made such observations, publishing what we call Goldich’s Weathering Series. The series ranked the ease with which common igneous minerals break down. Goldich found that minerals that crystallize from a magma at high temperature – minerals relatively poor in silicon and oxygen – are generally less resistant to weathering than those that crystallize at low temperature. Thus, the minerals at the top of Bowen’s reaction series weather most easily and those at the bottom are more resistant to weathering. Iron-magnesium silicates, such as olivine, pyroxene, or amphibole break down relatively easily. Calcic feldspars, and many minerals with high solubilities in water, are also quick to decompose. Quartz, some feldspars, and some non-silicate minerals are relatively resistant to weathering because they contain more bonds, especially Si – O bonds, that do not break easily. It should not be surprising that minerals that characterize high-temperature igneous rocks, or those most often dissolved in water, are the first to decompose under Earth surface conditions where temperature is low and water is abundant.
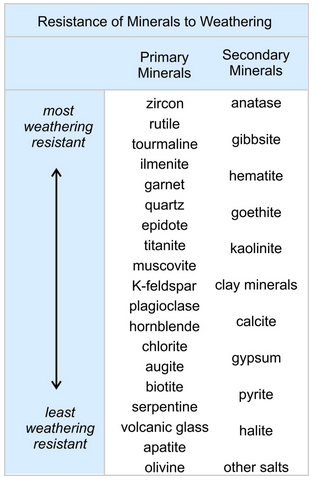
Sedimentologists have made comprehensive lists of the relative ease with which minerals weather. Although there is some variation, the list shown here is typical (from Birkeland 1999). Like primary minerals, secondary minerals can break down and disappear, so this table compares weathering rates for both primary minerals and secondary minerals. Weathering resistance, however, does not necessarily mean that a particular mineral is abundant in weathered materials. Some of the minerals at the top of the list in the table are uncommon compared with others. Zircon, rutile, and tourmaline, for example, are very resistant to weathering but rarely are major components of sediments because they are only minor minerals in most parent rocks. Minerals at the bottom of the list are very unstable when exposed to the elements and, consequently, are absent from all but the youngest sediments.
After chemical weathering, dissolved material is carried away. Residual minerals and secondary minerals such as clay may remain where they form. For example, prolonged weathering of bedrock can lead to thick layers of reddish soil called laterite in tropical areas. Laterites vary but are always rich in oxide minerals and clays. Laterites are easily eroded. Over time, erosion by water, gravity, or wind can transport laterite debris, just like any other detrital material, away from its place of origin.
Laterites and Bauxites
Consider a tropical area with warm weather and abundant rainfall. Weathering and leaching will be extreme, and even clay minerals may decompose. Normally soluble elements, and even relatively insoluble silica, will be dissolved and removed. The remaining material, called a residual deposit, is often composed primarily of aluminum oxides and hydroxides, the least soluble of all common minerals. We term such deposits laterites (if unconsolidated) or bauxites (if lithified into rock). Figure 7.7 shows bauxite in Australia.

Bauxites and laterites are our most important source of aluminum. But, the mineralogy of a laterite depends on the composition of rocks weathered to produce it. Laterites can also be important sources of iron, manganese, cobalt, and nickel, all of which have low solubilities in water.
Most laterites are aluminous. The most important aluminum ore (bauxite), is a mixture of several minerals, including the polymorphs boehmite and diaspore, both AlO(OH), and gibbsite, Al(OH)3. Bauxite is mined in large amounts in Australia and Indonesia, and in smaller quantities in the Americas and in Europe. In some places, relatively young laterites produce ore, but in Australia economical laterite deposits are more than 65 million years old.


