2.4.2: Covalent Bonds
- Page ID
- 18294
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Rather than giving up or gaining electrons to become ions, atoms with incompletely filled energy levels may become stabilized by sharing electrons. The sharing of pairs of electrons between atoms produces a covalent bond. If the sharing is complete, the bond is 100% covalent. This is the case for diatomic gases such as N2, but does not occur in common minerals except for diamond.
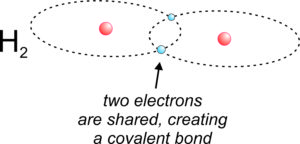
2.14 Covalent bonding to make hydrogen gas
The simplest example of a covalent bond involves two hydrogen atoms combining to produce H2 gas, shown in Figure 2.14. Hydrogen atoms have one proton in their nucleus and one electron in orbit. If two hydrogen atoms share electrons, this effectively puts two electrons in the outer orbits (1s) of each atom, completely filling the orbits and making a very stable diatomic molecule, H2. Fluorine, chlorine, and other halogen elements form covalently bonded diatomic gasses (F2, Cl2, etc.) like hydrogen.
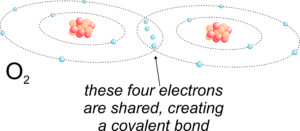
2.15 Covalent bonding to make oxygen gas
Sometimes covalent bonds involve more than a single pair of shared electrons. Figure 2.15 shows how two oxygen atoms can share four electrons to form covalent bonds, and produce O2 gas. By sharing four electrons, each atom has a total of 8 electrons in its 2s and 2p orbitals, making the outer shells fully occupied and stable. Because there are two pairs of electrons shared, we call this kind of covalent bond a double bond.
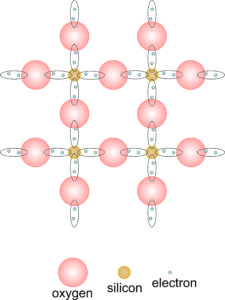
2.16 Simplified view of bonding in quartz
For a mineralogical example of covalent bonding, let’s consider quartz (SiO2). The drawing shown in Figure 2.16 is the atomic arrangement in quartz reduced to two dimensions. Four oxygen atoms surround each silicon atom, and each oxygen atom bonds to two silicon atoms. A lone silicon atom has 4 electrons in its outer shell (3s and 3p orbitals). But, each of the surrounding oxygen atoms can share an electron with the silicon atom, bringing the total number of electrons to 8, filling the outer shell, and making it stable. And, each silicon atom can share one electron with each of its four surrounding oxygen atoms. So, oxygen, which begins with 6 electrons in its outer level (2s and 2p orbitals), gains 2 electrons, bringing the total to 8, which stabilizes the atom.
Many elements form both covalent and ionic bonds. As mentioned previously, Cl– forms ionic bonds with Na+ to make the mineral halite. But, two Cl atoms can also bond covalently to form a stable gas, Cl2. The two types of bonds are not exclusive – ionic bonds and covalent bonds form a spectrum, and in nature, 100% ionic and covalent bonds do not exist. In nearly 100% ionic bonds, valence electrons are almost exclusively associated with one atom. (But, even the highly ionic NaCl bond has a slight covalent character.) In nearly 100% covalent bonds, electrons are shared more or less evenly between two atoms. Diatomic gases such as H2 and N2 are extremely close to being 100% covalent.
So, many minerals (and other substances) contain bonds that are partly ionic and partly covalent. For example, the preceding discussion of covalent bonding in quartz was an oversimplification. In actuality, silicon-oxygen bonds are not completely covalent – there is still some ionization and electrons are more localized around oxygen than silicon. The overall bonding is about half covalent and half ionic. And, although they behave as anions, (SiO4)2-, (SO4)2-, (CO3)2-, (OH)– and all other molecular ions (listed in the table above in Section 2.4.1.1) are held together by partially covalent bonds. This explains why they act as individual ionic units in ionic crystals.


