4.3: Chemistry of Minerals
- Page ID
- 33298
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Atoms
To understand mineral chemistry, it is essential to examine the fundamental unit of all matter, the atom.
Matter is made of atoms. Atoms consist of subatomic particles: protons, neutrons, and electrons. A simple model of the atom has a central nucleus composed of protons, which have a positive charge, and neutrons which have no charge. A cloud of negatively charged electrons surrounds the nucleus, with the number of electrons equaling the number of protons, balancing with the positive charge of the protons for a neutral atom. Protons and neutrons each have a mass number of 1. The mass of an electron is less than 1/1000th that of a proton or neutron, meaning most of the atom’s mass is in the nucleus. For our purposes, we can simplify the math by rounding the electron’s mass down to zero.
To summarize:
The subatomic particles in an atom are:
- protons - positively charged, mass = 1 amu, located in the nucleus
- neutrons - no charge, mass = 1 amu, located in the nucleus
- electrons - negatively charged, mass = 0 amu, located outside of the nucleus
This means that all of an atom's mass is in the nucleus. The charge of the atom depends on the balance of protons and electrons such that if the number of electrons equals the number of protons, the element has no charge.
The Periodic Table of Elements
Matter is composed of elements which are atoms that have a specific number of protons in the nucleus. This number of protons is called the atomic number of the element. For example, an oxygen atom has 8 protons and an iron atom has 26 protons. Down to the atom, each element retains unique chemical and physical properties, leading to particular behaviors in nature. This uniqueness led scientists to develop a periodic table of the elements, a tabular arrangement of all known elements listed in order of their atomic number and chemical properties.

The first arrangement of elements into a periodic table was done by Dmitri Mendeleev in 1869 using the elements known at the time [1]. In the periodic table, each element has a chemical symbol, name, atomic number, and atomic mass. The chemical symbol is an abbreviation for the element, often derived from a Latin or Greek name for the substance (for example, lead is Pb from the Latin ‘plumbum’) [2]. The atomic number is the number of protons in the nucleus. The atomic mass is the number of protons and neutrons in the nucleus, each with a mass number of one. Since the mass of electrons is so much less than that of protons and neutrons, the atomic mass is effectively the number of protons plus the number of neutrons. For example, the element silicon (Si) is atomic number 14 on the periodic table. Silicon has 14 protons and 14 neutrons in its nucleus and 14 electrons located in its electron cloud. The atomic mass of silicon is 28.01 mass units, with the 0.01 being the mass of the electrons.
To summarize:
Just like there are brands of cars there are elements of atoms. So Toyota and Honda are brands (or a type of car), just like Oxygen and Silicon are elements (or a type of atom). The element an atom is, is based on the number of protons. Oxygen is always 8; Silicon is always 14. To find the mass of an atom you add the number of protons and neutrons (since electrons carry negligible mass). Silicon is therefore 28 (14 protons + 14 neutrons).
Isotopes
Atoms of the same element can have differing numbers of neutrons in their nucleus. These are referred to as isotopes of that element. Isotopes behave the same chemically however they are slightly different in mass.
Isotopes are very important to geology. Later you see how they are used to give ages to certain rocks.
To summarize and give an example:
To add to our car analogy from above, isotopes are varieties of elements just like there are different models of cars. For example Honda makes many models including Accords and Civics. Oxygen has several isotopes as the number of protons remains at 8 (that is what makes it Oxygen) and the number of neutrons varies between 8, 9, and 10. Accordingly the mass of the isotopes varies as follows:
- Oxygen isotope with 8 neutrons has a mass of 16 (8 protons + 8 neutrons); we call this O-16
- Oxygen isotope with 9 neutrons has a mass of 17 (8 protons + 9 neutrons); we call this O-17
- Oxygen isotope with 10 neutrons has a mass of 18 (8 protons + 10 neutrons); we call this O-18
Common Elements of the Earth’s Crust
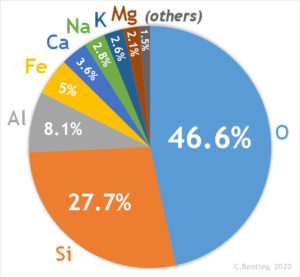
Among the 118 known elements appearing on the periodic table, only eight are abundant in the Earth’s crust and are shown in the pie chart below. Therefore, it is of these eight elements that the most common rock-forming minerals are composed.
Chemical Bonding
Minerals are formed by the chemical bonding that occurs between these elements. Most minerals are compounds containing multiple elements bonded together in a specific arrangement. Chemical bonding describes how these atoms attach with each other to form compounds. Halite (table salt) forms as sodium (Na) and chlorine (Cl) combine with the chemical formula: NaCl. There are some minerals that are not compounds as they are composed of only one element. Examples include native Copper (Cu), native Gold (Au), or native Silver (Ag). These single elements bond to each other to form molecules.
Valence, Charge and Ions
The electrons around the atom’s nucleus are located in shells representing different energy levels. The outermost shell is called the valence shell and the electrons here are involved in chemical bonding. Atoms are more stable when this valence shell is full, so most elements tend to gain or lose electrons to achieve this. If an atom gains or loses electrons in its valence shell, the number of protons and electrons are no longer equal and it gains an either positive or negative charge. This charged atom is called an ion. If protons outnumber electrons the charge is positive and it is called a cation. If electrons outnumber protons the charge is negative and it is called an anion.
Let’s use a familiar example to help illustrate. Sodium (Na) is located on the far left side of the Periodic Table. It has one lonely electron in its valence shell that it desperately wants to shed. Currently, one atom of sodium (Na) has an equal number of protons and electrons so it has no change. When it loses the one electron, it becomes positively charged (more protons than electrons) and is a cation designated as \(\ce{Na^+}\). Coincidentally, chlorine (Cl) has one spot left for an electron in its valence shell. Chlorine (Cl) gains an electron, and becomes negatively charged (more electrons than protons) and is an anion designated as \(\ce{Cl^-}\).
To summarize:
- ions form as atoms lose or gain electrons resulting in a positively or negatively charged atom
- ions that lose electrons have more protons than electrons and have a positive charge; they are called cations
- ions that gain electrons have more electrons than protons and have a negative charge; they are called anions
A chemical bond form between these oppositely charged ions which creates a molecule. This type of chemical bond is an ionic bond where electron transfer and a strong attraction between oppositely charged ions creates the bond. If this happens repeatedly, the mineral halite forms and is known as NaCl.


Explore the following 3D molecular crystal structure of the mineral halite, below. Roll your cursor over to grab and rotate the structure. The grey dots are Na atoms and the green dots are Cl atoms.
Now, take a look at a 3D representation of perfect mineral crystals of halite.
Do you notice the similarity of the molecular structure with the outward appearance of the crystals? The outward physical appearance of a mineral will reflect its internal atomic structure. How cool is it that the atomic level structure of a mineral is revealed in its shape! We will look at this in more detail later.
Another type of chemical bonding can occur between elements in the formation of minerals that involves electron sharing. This is most easily explained in looking at a simple molecular model of \(\ce{H2O}\), water. Oxygen has 6 electrons in its valence shell and hydrogen has only one. Oxygen is missing 2 electrons to be stable (that shell wants 8 electrons) and hydrogen is missing one electron (that shell wants 2 electrons). Having two hydrogen atoms and one oxygen atom share their electrons, the valence shells will contain 8 electrons for oxygen and 2 for the hydrogens. Both configurations are very stable. This type of bond is called a covalent bond and is stronger than an ionic bond.
The Silica Tetrahedron
The most common covalent bond in the formation of minerals is the bond that occurs between silicon and oxygen which are the two most common elements in the Earth’s crust. It makes sense that the most common minerals in the crust will therefore contain a lot of these two elements. The first nine of “The Big Ten” minerals are indeed from the largest mineral group, the silicate minerals. The silicates are named for the silicon and oxygen foundation in their composition. The basic framework of the silicate minerals is the silicon - oxygen tetrahedron (or silica tetrahedron) where four oxygen atoms pack very tightly around one silicon atom. The resulting molecular structure is shaped like a tetrahedron.

Since the the overall charge of the silica tetrahedra is -4 the molecule acts as an anion. As a result, this silica molecule is ready to bond with other available elements or complex molecules. This is how all of the silicate minerals form.
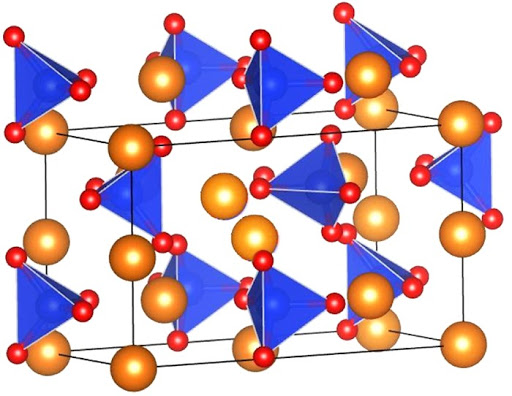
Let’s look at one of our “Big Ten” silicate minerals, olivine, to see how this works.
Olivine most commonly forms as a magma cools and minerals begin to crystallize. The chemical formula for the mineral olivine is: \(\bf{\ce{(Mg,Fe)2SiO4}}\) The elements of Mg (magnesium) and Fe (iron) appear together in parentheses because they can readily substitute for each other in the mineral’s crystalline structure depending on which of the elements are available in the cooling magma.

Both Mg and Fe have a valence charge of +2; both Mg and Fe have two electrons in their outermost shells, e.g. electrons that they are looking to “give away.” They are swimming around in that magma looking for partners! As the magma cools, it will reach a temperature at which bonding can occur. As discussed, the complex silica molecule forms with the overall -4 charge. The available Fe+2 and Mg+2 will form an ionic bond with the complex silica molecule which will balance out the overall charge.
- anion - a negatively charged ion formed as the number of electrons exceeds the number of protons
- atomic mass - the mass of an atom determined by adding the number of protons and neutrons in the atom
- atomic number - the number of protons in an atom which defines what element the atom is
- cation - a positively charged ion formed as the number of protons exceeds the number of electrons
- covalent bond - a strong type of bond in which atoms share electrons to complete their valence shells rending them stable
- electron - a negatively charged subatomic particle with essentially no mass that resides outside of an atom's nucleus
- ionic bond - a type of bond in which atoms lose or gain electrons to become negatively or positively charged and thus attracted to one another
- isotopes - varieties of a given element that vary in their number of neutrons and therefore in their atomic mass
- neutron - a subatomic particle with no charge having a mass of 1 atomic mass unit that resides inside an atom's nucleus
- nucleus - the central portion of an atom in which protons and neutrons reside
- proton - a positively charged subatomic particle having a mass of 1 atomic mass unit that resides inside an atom's nucleus
- silica tetrahedron - a molecule composed of one silicon atom and four oxygen atoms arranged in a tetrahedron with the silicon in the middle
[1] Mendeleev, D. The relation between the properties and atomic weights of the elements. Journal of the Russian Chemical Society 1, 60–77 (1869)
[2] Scerri, E. R. The Periodic Table: Its Story and Its Significance. (Oxford University Press, USA, 2007)


