6.2: Metamorphic Processes
- Page ID
- 6874
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Metamorphism occurs when solid rock changes in composition and/or texture without the mineral crystals melting, which is how igneous rock is generated. Metamorphic source rocks, the rocks that experience the metamorphism, are called the parent rock or protolith, from proto– meaning first, and lithos- meaning rock. Most metamorphic processes take place deep underground, inside the earth’s crust. During metamorphism, protolith chemistry is mildly changed by increased temperature (heat), a type of pressure called confining pressure, and/or chemically reactive fluids. Rock texture is changed by heat, confining pressure, and a type of pressure called directed stress.
Temperature (Heat)
Temperature measures a substance’s energy—an increase in temperature represents an increase in energy [2]. Temperature changes affect the chemical equilibrium or cation balance in minerals. At high temperatures, atoms may vibrate so vigorously they jump from one position to another within the crystal lattice, which remains intact. In other words, this atom swapping can happen while the rock is still solid.
The temperatures of metamorphic rock lie in between surficial processes (as in sedimentary rock) and magma in the rock cycle. Heat-driven metamorphism begins at temperatures as cold as 200˚C and can continue to occur at temperatures as high as 700°C-1,100°C [3; 4; 5]. Higher temperatures would create magma, and thus, would no longer be a metamorphic process. Temperature increases with increasing depth in the Earth along a geothermal gradient (see Chapter 4) and metamorphic rock records these depth-related temperature changes.
Pressure
Pressure is the force exerted over a unit area on a material. Like heat, pressure can affect the chemical equilibrium of minerals in a rock. The pressure that affects metamorphic rocks can be grouped into confining pressure and directed stress. Stress is a scientific term indicating a force. Strain is the result of this stress, including metamorphic changes within minerals.
Confining Pressure
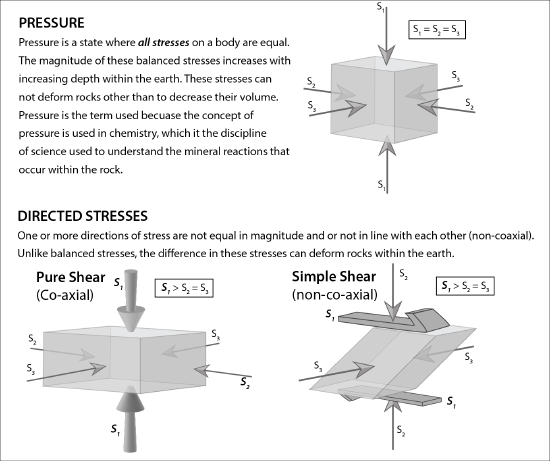
Pressure exerted on rocks under the surface is due to the simple fact that rocks lie on top of one another. When pressure is exerted from rocks above, it is balanced from below and sides and is called confining or lithostatic pressure. Confining pressure has equal pressure on all sides (see figure) and is responsible for causing chemical reactions to occur, just like heat. These chemical reactions will cause new minerals to form.
Confining pressure is measured in bars and ranges from 1 bar at sea level to around 10,000 bars at the base of the crust [6]. For metamorphic rocks, pressures range from a relatively low-pressure of 3,000 bars [5] around 50,000 bars [5], which occurs around 15-35 kilometers below the surface.
Directed Stress

Directed stress, also called differential or tectonic stress, is an unequal balance of forces on a rock in one or more directions (see the previous figure). Directed stresses are generated by the movement of lithospheric plates. Stress indicates a type of force acting on the rock. Strain describes the resultant processes caused by stress and includes metamorphic changes in the minerals. In contrast to confining pressure, directed stress occurs at much lower pressures and does not generate chemical reactions that change the mineral composition and atomic structure [3]. Instead, directed stress modifies the parent rock at a mechanical level, changing the arrangement, size, and/or shape of the mineral crystals. These crystalline changes create identifying textures, which is shown in the figure below comparing the phaneritic texture of igneous granite with the foliated texture of metamorphic gneiss.

Directed stresses produce rock textures in many ways. Crystals are rotated, changing their orientation in space. Crystals can get fractured, reducing their grain size. Conversely, they may grow larger as atoms migrate. Crystal shapes also become deformed. These mechanical changes occur via recrystallization, which is when minerals dissolve from an area of rock experiencing high stress and precipitate or regrow in a location having lower stress. For example, recrystallization increases grain size much like adjacent soap bubbles coalesce to form larger ones. Recrystallization rearranges mineral crystals without fracturing the rock structure, deforming the rock-like silly putty; these changes provide important clues to understanding the creation and movement of deep underground rock faults.
Fluids
A third metamorphic agent is chemically reactive fluids that are expelled by crystallizing magma and created by metamorphic reactions. These reactive fluids are made of mostly water (H2O) and carbon dioxide (CO2), and smaller amounts of potassium (K), sodium (Na), iron (Fe), magnesium (Mg), calcium (Ca), and aluminum (Al). These fluids react with minerals in the protolith, changing its chemical equilibrium and mineral composition, in a process similar to the reactions driven by heat and pressure. In addition to using elements found in the protolith, the chemical reaction may incorporate substances contributed by the fluids to create new minerals. In general, this style of metamorphism, in which fluids play an important role, is called hydrothermal metamorphism or hydrothermal alteration. Water actively participates in chemical reactions and allows extra mobility of the components in hydrothermal alteration.
Fluids-activated metamorphism is frequently involved in creating economically important mineral deposits that are located next to igneous intrusions of magma bodies. For example, the mining districts in the Cottonwood Canyons and Mineral Basin of northern Utah produce valuable ores such as argentite (silver sulfide), galena (lead sulfide), and chalcopyrite (copper-iron sulfide), as well as the native element gold [2]. These mineral deposits were created from the interaction between a granitic intrusion called the Little Cottonwood Stock and country-rock consisting of mostly limestone and dolostone. Hot, circulating fluids expelled by the crystallizing granite reacted with and dissolved the surrounding limestone and dolostone, precipitating out new minerals created by the chemical reaction. Hydrothermal alteration of mafic mantle rock, such as olivine and basalt, creates the metamorphic rock serpentinite, a member of the serpentine subgroup of minerals. This metamorphic process happens at mid-ocean spreading centers where newly formed oceanic crust interacts with seawater.
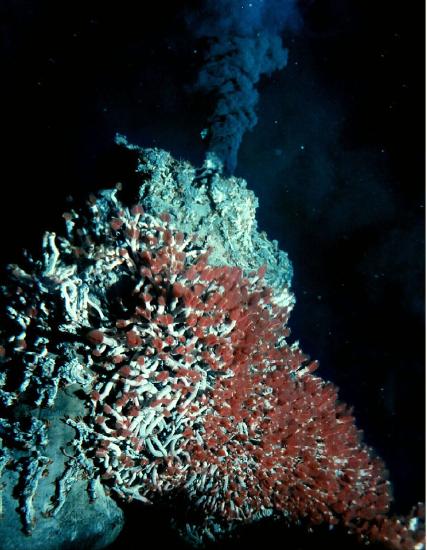
Some hydrothermal alterations remove elements from the parent rock rather than deposit them. This happens when seawater circulates down through fractures in the fresh, still-hot basalt, reacting with and removing mineral ions from it. The dissolved minerals are usually ions that do not fit snugly in the silicate crystal structure, such as copper. The mineral-laden water emerges from the seafloor via hydrothermal vents called black smokers, named after the dark-colored precipitates produced when the hot vent water meets cold seawater. (see Chapter 4, Igneous Rock and Volcanic Processes) Ancient black smokers were an important source of copper ore for the inhabitants of Cyprus (Cypriots) as early as 4,000 BCE, and later by the Romans [8].
References
- 2. Jeong I-K, Heffner RH, Graf MJ, Billinge SJL (2003) Lattice dynamics and correlated atomic motion from the atomic pair distribution function. Phys Rev B Condens Matter 67:104301
- 3. Winter JD (2009) Principles of igneous and metamorphic petrology, 2 edition. Pearson, New York
- 4. Boehler R (1996) Melting temperatures of the Earth’s mantle and core: Earth’s thermal structure. Annual Review of Earth and Planetary Sciences 24:15–40. https://doi.org/10.1146/annurev.earth.24.1.15
- 5. Issitt ML (2012) Ultra-High-Pressure Metamorphism. Earth Science: Earth Materials & Resources 523
- 6. Petrini, Podladchikov (2000) Lithospheric pressure–depth relationship in compressive regions of thickened crust. Journal of Metamorphic Geology 18:67–77. https://doi.org/10.1046/j.1525-1314.2000.00240.x
- 8. (2007) Timeline of Art History. Reference Reviews 21:45–45


