17.18: Toxicity
- Page ID
- 51550
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Essential to Know
- A chemical is toxic if it can cause death or adverse sublethal effects in organisms exposed to it at a concentration above a critical threshold.
- Chemicals that are toxic can have many possible sublethal adverse effects on organisms, such as inhibition of the ability to photosynthesize or feed. The most important sublethal effects appear to be those that interfere with reproductive success.
- All chemicals that can be toxic have a concentration threshold below which they have no sublethal or lethal toxic effects. Hence, for every chemical in the ocean environment, there is a concentration below which it is environmentally safe.
- Many substances that are toxic at high concentrations are also essential to life, and the growth of marine organisms may be inhibited if they are not present at concentrations above a certain threshold. For these substances, there is an optimum range between the minimum concentration that an organism requires to meet its needs and the concentration above which the substance is toxic.
- The range of optimum concentration can be large or small. The concentration at which toxicity occurs can be many times higher than or very close to the natural range.
- Anthropogenic inputs of potentially toxic substances can be assimilated safely in the oceans if the amount introduced does not cause concentrations to exceed the threshold at which sublethal toxicity occurs. The quantity that can be safely assimilated is different for each substance and is determined in part by its sublethal toxicity threshold and background concentrations.
- Sublethal or lethal toxicity threshold concentrations are difficult to determine because they vary among species, among substances, and with other factors, such as physical stresses and synergistic and antagonistic effects of other chemical constituents.
- Marine organisms bioaccumulate most toxic substances. Bioaccumulation occurs when the concentration in the organism is higher than the environmental concentration, but the concentrations are in equilibrium.
- A few toxic substances are biomagnified in marine organisms. Biomagnification occurs when the organism retains all the toxic substance to which it is exposed in its food or environment and does not lose any of the substance, even if its environmental concentration decreases.
- Carcinogenicity, mutagenicity, and teratogenicity can be considered to be lethal or sublethal effects, but there is probably no concentration threshold below which there is no effect. Each exposed organism has a small probability of suffering an effect at any specific concentration. More individuals within a population suffer the effects as the concentration increases.
- Carcinogenic, mutagenic, and teratogenic substances occur naturally. Anthropogenic inputs will increase the incidence of the effects of such substances. However, at least for some of these substances, anthropogenic inputs would need to be large before the increased incidence would be significant or measurable in comparison with the natural incidence of effects.
Understanding the Concept
A chemical is toxic if it is present at high enough concentrations to cause death or adverse sublethal effects in organisms, including human beings, that are exposed to the substance. If an organism is exposed to a concentration that is lethally toxic, the organism dies. If an organism is exposed to a concentration that is sublethally toxic, the organism is not killed, but it is disadvantaged in some way.
Examples of the many possible sublethal effects are partial inhibition of photosynthesis in plants and partial inhibition of feeding, hunting, or prey avoidance capabilities in animals. However, the most important sublethal effects are generally believed to be those that reduce reproductive success. Reproductive success can be reduced by partial inhibition of egg or sperm production, reduction in the probability of fertilization when egg and sperm meet, reduction in the survival ability of larvae or juveniles, and in other ways. Because of the wide range of possible sublethal effects caused by toxic chemicals, identifying and measuring such effects is often impossible either in the environment or in laboratory experiments.
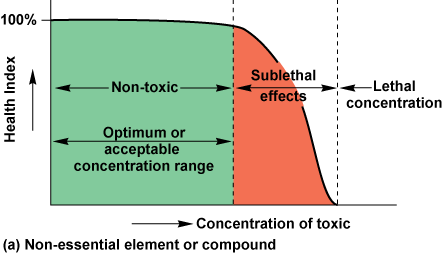
The effects of any chemical that is potentially toxic vary with its concentration (Fig. CC18-1
All substances have an acceptable or optimum concentration range within which they do not harm organisms or the ecosystem. Therefore, anthropogenic additions of chemicals to the oceans are not always harmful. Such inputs can be safe if the assimilative capacity or acceptable concentration range in the environment is not exceeded. The assimilative capacity is determined by the sublethal toxicity threshold of the most sensitive species.
The relationships shown in Figure CC18-1 generally apply to all chemicals other than carcinogens, mutagens, and teratogens. However, concentrations at which the inhibition of growth ends (for essential nutrients), sublethal toxicity begins, and lethal toxicity occurs can all vary by orders of magnitude for different species, and even for different life stages of a species, exposed to a specific substance.
Most importantly, the optimum concentration range for essential nutrients may vary dramatically. For example, zinc is a toxic but essential chemical that has a wide optimum concentration range. In certain parts of the oceans, zinc may be the nutrient most likely to be depleted and hence most likely to restrict the growth of certain organisms. Because it has a large optimum concentration range, zinc becomes sublethally toxic only at concentrations that are many orders of magnitude above those normally present in the oceans. In contrast, copper has a very narrow optimum concentration range. Like zinc, copper may be growth-limiting to certain species in some parts of the oceans. However, at concentrations only a few times higher than the limiting concentration and within the range often present in contaminated coastal waters, copper can reduce the rate of photosynthesis by certain species of phytoplankton. Hence, copper is sublethally toxic at concentrations very close to those that occur naturally in the oceans.
Acceptable or optimum concentration ranges and the range of concentrations present naturally must both be considered in assessments of whether anthropogenic discharges might create pollution problems. For example, much smaller quantities of copper than of zinc can be discharged safely to the oceans.
Concentrations that represent the transitions between growth inhibition, nontoxicity, sublethal toxicity, and lethal toxicity are very difficult to determine for several reasons. First, members of a species differ somewhat in susceptibility to a given toxic chemical. Second, different species and life stages of a single species may have dramatically different susceptibilities to a particular chemical. Third, complicated, antagonistic, and synergistic effects occur among potentially toxic substances and other chemicals in the environment. Synergistic effects increase toxicity. For example, high concentrations of one trace metal can increase the toxicity of another trace metal. Antagonistic effects are the opposite; high concentrations of one trace metal may reduce the toxicity of another metal. Fourth, the susceptibility of a species to a chemical may be altered by stress due to other environmental factors, including temperature, salinity, and the extent of competition from other species. Fifth, the toxicity of many substances is determined by their chemical form. For example, ionic copper is substantially more toxic than copper complexed with organic matter.
Because of the variability among chemicals, among species, among species’ life stages, among chemical states, and with synergistic, antagonistic, and stress factors, it is not possible to determine a single concentration at which a chemical is lethal or sublethal in the environment. For this reason, toxicologists often measure lethal toxic concentrations of chemicals on selected species in the laboratory by using bioassays. They assume that sublethal effects will occur at concentrations below the lethal concentration. They also assume that some species are more sensitive to the toxic chemical than the tested species is. A “safe” concentration in the environment is then estimated. Generally, a concentration two orders of magnitude below the minimum lethal concentration is arbitrarily assumed to be safely below the level at which any sublethal adverse effects occur. However, in some cases, such as copper, the optimum concentration range (Fig. CC18-1b) is less than two orders of magnitude below the lethal concentration threshold. Therefore, this approach may set the “safe” concentration below naturally occurring levels and sometimes even below the level needed to sustain growth.
Bioassays are conceptually simple. The test species is usually exposed for 96 h (a duration chosen because one set of tests can be set up and run in a 5-day workweek) to several different concentrations of the potentially toxic substances in seawater. The test species is also exposed to control seawater with none of the potentially toxic substances added. After exposure, the number of organisms that have died or exhibited a sublethal effect (e.g., failure of an embryo to develop or of a larval stage to metamorphose to the next life stage) are counted. Generally, no death or sublethal effect occurs in the control seawater (otherwise the experiment is not acceptable, because a factor other than the tested contaminant has caused a confounding effect). If the test substance is toxic, some or all individuals will have died or shown the sublethal effect in the test with the highest contaminant concentration. A progressively smaller proportion of the test population is usually affected at lower contaminant concentrations. From these tests, the contaminant concentrations at which one-half of the test organisms are killed or suffer the observed sublethal effect are calculated. These values are called the LC50 and EC50, respectively.
Bioassay tests must be interpreted carefully because they do not use the most sensitive organism in the ecosystem being assayed. Without testing all species, there is no way to determine which is the most sensitive. In fact, because different life stages of some species have different levels of sensitivity (juvenile stages are generally more sensitive than adults), all life stages of all species would have to be tested. In addition, most marine species are difficult to keep alive in the laboratory because this alien environment stresses the organisms. Species used for bioassays must be stress-tolerant, and hence they are also likely to be tolerant of toxicant stress.
Many factors, such as salinity, temperature, light intensity, water chemistry, sediment characteristics, food supply, and competition or cooperation with other individuals or species, are different in bioassays than in the environment. The additional stresses of the test environment may make test organisms more susceptible to toxicant stress.
Bioassays are often conducted with samples of wastes in the form discharged (e.g., sewage or industrial effluent). Although this approach takes into account synergism or antagonism between components of the waste, there remains the question of whether additional such effects occur when the waste is mixed in seawater and subject to different conditions in the environment.
Most chemicals that exhibit toxicity are more highly concentrated in tissues of marine species than in the surrounding seawater. This fact has led to a popular belief that toxic chemicals are taken up continuously by marine species and that concentrations of the toxins in their tissues increase progressively throughout their lifetime, regardless of how the concentration changes in their environment. This may be true for some species and some chemicals, but it is not true for most species and most substances. Most potentially toxic chemicals are bioaccumulated by most marine species, but are not biomagnified.
Bioaccumulation occurs when the concentration of a compound is regulated by equilibria between the organism and its food and/or surrounding water. The substance is taken in by the organism from its food and/or directly from the water, but the organism is capable of excreting some of the substance either directly back to the water or through its urine or feces. Hence, if the food or seawater concentration of the substance increases, the concentration in the organism will increase, but if the food or seawater concentration decreases, the concentration in the organism will also decrease. However, it may take some time for equilibrium to be reached.
Certain toxic substances are more dangerous than others in marine ecosystems because they biomagnify. Biomagnification occurs when organisms at each trophic level in a food chain retain all or almost all of a toxic substance ingested in food (CC15). Consequently, the concentration of the toxin increases at each trophic level.
Certain chemicals are carcinogenic (cancer-causing), mutagenic (causing genetic changes in the offspring by altering the parental DNA), or teratogenic (causing abnormal development of the embryo). Although each can be considered a sublethal effect, many scientists do not believe that these effects follow the toxicity-concentration relationships shown in Figure CC18-1, but rather that they may occur at any concentration of these substances.
If this hypothesis is correct, a single molecule of such a chemical could cause cancer in an exposed individual. However, if many individuals were each exposed to a single molecule, only a few (if any) would be expected to develop cancer. If the same group of individuals were each exposed to two molecules of the chemical, twice as many would be expected to develop cancer. This type of effect is said to be “probabilistic.” The probability that any one individual will contract cancer from a given concentration of the chemical is reduced proportionally as the concentration is reduced, but it never reaches zero.
There are many naturally occurring carcinogens, teratogens, and mutagens, but only a very small percentage of marine populations suffer adverse effects as a result of these naturally occurring compounds. Because such a small fraction is affected, great numbers of individuals would have to be monitored to accurately estimate the percentage of a population that suffers such effects. Furthermore, determining the extent to which a higher incidence of such effects might be caused by human contamination of the oceans, or parts thereof, would be even more difficult.
If the probabilistic-effect hypothesis is correct, any quantity of anthropogenic input of a carcinogen, mutagen, or teratogen will increase the incidence of such effects in marine species. The increase may be extremely small in comparison with the incidence of such effects due to the naturally occurring concentrations, in which case many scientists believe adverse effects on the marine ecosystem would be negligible. Hence, some believe that it would be environmentally safe and acceptable to dispose of limited quantities of anthropogenic carcinogens, mutagens, and teratogens in the ocean.