17.14: Phototrophy, Light, and Nutrients
- Page ID
- 51546
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Essential to Know
- Organic matter is created from inorganic substances (primary production) by phototrophy and chemosynthesis.
- Phototrophy is performed by organisms that use light as their energy source to fuel the primary production process. There are two known mechanisms: photosynthesis and a recently discovered metabolic process that uses compounds called rhodopsins to capture light energy.
- Most primary production in Earth’s ecosystems is created by oxygenic photosynthesis, but too little is known about the use of the rhodopsin-based mechanism in ocean ecosystems to assess its relative importance.
- Plants and algae are eukaryotes that perform oxygenic photosynthesis through a complex series of reactions that occur in pigmented cells called “chloroplasts.”
- Photosynthesis originated and still takes place in certain Prokaryotes called Cyanobacteria. Chloroplasts are thought to have evolved from photosynthetic symbiotic cyanobacteria that became a permanent resident in a eukaryotic cell.
- A nitrogen-containing pigment called “chlorophyll a” plays the central role in transferring light energy to the oxygenic photosynthesis process. Chloroplasts contain accessory pigments that capture light energy and transfer it to chlorophyll a.
- Accessory pigments collect light energy at wavelengths at which chlorophyll a absorbs light poorly. Each species has a different suite of accessory pigments optimized to collect light energy available in its specific habitat.
- Oxygenic photosynthesis uses light energy, carbon dioxide, and water to produce oxygen (which is released) and relatively simple organic compounds. Most organic compounds produced by oxygenic photosynthesis are used for cellular respiration. Only a small proportion enters biochemical cycles and is converted to more complex organic molecules.
- Only a very small percentage of the light energy used in photosynthesis is used to produce biomass.
- Nitrogen and phosphorus, among other elements, are present in key compounds involved in photosynthesis. Hence, depletion of available nitrogen and phosphorus stops production of these compounds and limits primary productivity.
- Anoxygenic photosynthesis is an alternate pathway employed by anaerobic bacteria. Anoxygenic photosynthesis uses light energy, carbon dioxide, and a hydrogen donor, such as H2S or H2, to create relatively simple organic compounds and produces a very small amount of organic matter in comparison with algal and cyanobacterial photosynthesis in most present-day ocean ecosystems.
- Chemosynthetic bacteria and archaea synthesize organic compounds by using chemical energy from the oxidation of compounds such as H2S and H2 instead of light energy.
Understanding the Concept
For life to exist, carbon, hydrogen, oxygen, nitrogen, phosphorus, and many other essential elements must be synthesized into organic compounds. It is believed that most organic matter on the Earth today was initially converted from inorganic matter to organic compounds by photosynthesis. Exceptions include a few simple organic compounds created by electrical discharges in the atmosphere, and the chemosynthetic communities described in Chapters 12 and 15, the importance of rhodopsin-based phototrophy is uncertain.
Photosynthesis is a complex process in which plants, algae, and certain bacteria and archaea use light energy to convert carbon dioxide into organic compounds. There are two major known photosynthetic pathways: oxygenic and anoxygenic photosynthesis. There are also variations of these pathways, such as those used by certain archaea. Because their photosynthetic mechanisms differ from those of plants and algae, organisms that use these variations are often not considered to use photosynthesis by biologists who prefer to restrict the definition of photosynthesis to the specific pathway used in plants. In this text, we use a broader definition of photosynthesis that includes all pathways used by any organisms that use chlorophylls to capture light energy and carbon dioxide to produce organic matter. Organisms that use light energy to provide sufficient organic matter to generate new biomass are called phototrophs or photoautotrophs.
The first step in all variations of photosynthesis is the capture of light energy by the photosynthetic organism. In oxygenic photosynthesis by plants and algae, light is captured in complex cell structures called “chloroplasts” located near the organism’s surface. The chloroplasts contain chlorophyll a and, in many species, the related pigments chlorophyll b and chlorophyll c. There are also a number of “accessory pigments,” including carotenoids, xanthophylls, phycobilins, and chlorophyll d and chlorophyll f. Each photosynthetic species has its own unique combination of pigments. Accessory pigments capture light energy and transfer it to chlorophyll a, which then performs the next step in the synthesis. Chlorophylls b and c can be characterized as accessory pigments because they, too, collect light energy and pass it on to chlorophyll a. The transfer of energy from accessory pigments to chlorophyll a is an extremely efficient process. In some cases, transfer efficiency approaches 100%.
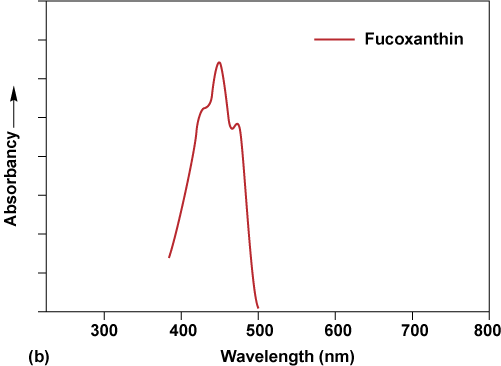
Why are accessory pigments needed? Chlorophyll a absorbs light effectively only toward the red and violet ends of the spectrum (Fig. CC14-1a). To capture light energy from the central blue–green–yellow part of the spectrum, other pigments are needed. Carotenoids, which include the group of pigments called xanthophylls, absorb energy primarily in the blue and blue-green parts of the spectrum (Fig. CC14-1b), whereas phycobilins absorb energy primarily in the green, yellow, and ultraviolet parts (Fig. CC14-1c). Chlorophylls b and c also absorb in different parts of the visible spectrum than chlorophyll a (Fig. CC14-1a). Chlorophyll d and chlorophyll f absorb in the infrared wavelengths. Thus, the chloroplast contains an array of accessory pigments to capture as much of the available light energy as possible. Accessory pigments transfer light energy that they capture to chlorophyll a, which performs the first steps in photosynthesis.
Red and blue-violet light are absorbed more effectively by seawater than are green and blue light (Fig. 5-17). Hence, accessory pigments are especially important in marine algae that must photosynthesize below the immediate surface layer. Absorption spectra of seawater in the open ocean and coastal waters are different (Fig. 5-18), and the wavelength distribution of light changes with depth. Species of marine algae that live at different locations and depths have different accessory pigment compositions to optimize their capture of available light energy.
Photosynthetic cyanobacteria also use chlorophyll or a closely related pigment, bacteriochlorophyll, to perform oxygenic photosynthesis, but they do not have chloroplasts. Photosynthesis is believed to have originated in cyanobacteria and, over a period of about 1 billion years. They transformed Earth’s atmosphere from one with almost no oxygen to the oxygen-rich atmosphere that now exists. Eukaryotes developed only toward the end of this transformation process, and chloroplasts are thought to have originated from a symbiotic cyanobacteria that became permanently located within its host Eukaryote cells.
Some archaea (and perhaps bacteria) use a complex protein, such as archaerhodopsin or halorhodopsin, to collect light energy and produce organic compounds for their metabolic needs, so these organisms employ phototrophy, but they are not photosynthesizers. At present, it is not known how important this rhodopsin-based phototrophy is in the overall primary production in the oceans, but its contribution is thought to be comparatively small. In contrast, cyanobacteria are estimated to produce about one-half of all ocean primary production. They do so because they appear to be the principal primary producers in the large areas of the oceans that are nutrient-limited. Thus, although their population and primary productivity per unit area of oceans are low, their total numbers and total primary production rivals the total primary production by algae and plants in the ocean.
The key molecule in oxygenated photosynthesis, chlorophyll a (Fig. CC14-2), is a complex molecule that contains not only carbon (C), hydrogen (H), and oxygen (O) atoms, but also a magnesium atom (Mg) and, most importantly, nitrogen atoms (N). The other forms of chlorophyll and rhodopsins also contain nitrogen. When dissolved nitrogen is depleted in ocean waters, nitrogen-containing compounds, including chlorophyll a, cannot be synthesized by algae and primary production is limited or stopped (Chap. 12). Thus, nitrogen as nitrate (NO3–), nitrite (NO2–), or ammonia (ammonium ion [NH4+] and NH3) is essential to photosynthesis. The exception to this is that certain cyanobacteria can convert molecular nitrogen to ammonia, although the conversion process is energy intensive and takes place very slowly, especially in oxygenated environments such as most ocean water.

After chlorophyll a has captured light energy, it can transfer an excited electron (an electron to which excess energy has been added) to other molecules, which transfer the energy through yet other molecules in a complex series of steps. During these transfers, the raw materials of photosynthesis—water and carbon dioxide—are brought together and converted into organic compounds.
Some reactions in photosynthesis occur only in the presence of light. The complex process of oxygenated photosynthesis can be simplified and summarized as follows. Light energy is used to split a water molecule into a hydrogen atom with an excited electron and a hydroxyl ion, each of which immediately reacts with other molecules in the chloroplast. The hydroxyl ion is then converted to oxygen and water, which are released, and the hydrogen atom with its excited electron is combined with NADP (nicotinamide adenine dinucleotide phosphate) to form NADP-H. At the same time, additional light energy is transferred to ADP (adenosine diphosphate) to form ATP (adenosine triphosphate) (Fig. CC14-3). ATP is a versatile energy-storing molecule that can donate the considerable energy associated with one of its phosphate bonds by releasing or transferring this phosphate group, which changes ATP back into ADP. ATP is an energy provider in both photosynthesis and respiration. Both ATP and ADP contain phosphorus, which is therefore also an essential nutrient for photosynthesis.

Once light energy is stored in ATP and NADP-H, the remaining steps in photosynthesis can continue in the dark. However, if light is removed for an extended period, the store of these molecules in the chloroplast is used up. The photosynthesis steps that can occur in the dark use hydrogen from NADP-H and energy from ATP to reduce carbon dioxide. Oxygen is removed from carbon dioxide and released, whereas the carbon is combined with other carbon atoms to form organic compounds.
There are several different pathways for these reactions, each producing a different organic compound or compounds. One of the most common pathways leads to the production of glucose (C6H12O6). Glucose and other organic molecules formed by photosynthesis are subsequently used by the organism as basic building blocks for the vast array of other organic compounds produced by living organisms in their biochemical cycles. Large amounts of glucose and the other compounds created by photosynthesis are also converted back into carbon dioxide in the respiration processes of the plant. The overall efficiency of transferring light energy through photosynthesis to create new biomass is extremely low, a few percent at most.
The process of oxygenic primary production can be depicted by the following, much simplified equation:
6CO2 + 6H2O →visible light→ C6H12O6 + 6O2
Cyanobacteria photosynthesize in a somewhat different way. First, cyanobacteria contain a unique form of chlorophyll called “bacteriochlorophyll.” In addition, they do not use water to provide hydrogen for photosynthesis, and they do not release oxygen. Cyanobacterial photosynthesis can be depicted by the following simplified equation:
6CO2 + 6 H2[A] →visible and far red light→ C6H12O6 + 6[A]
H2[A] is a hydrogen donor and can be H2S, H2, or various organic compounds. Cyanobacterial photosynthesis does not make a major contribution to ocean primary production in most ocean areas, because environments where both light and a suitable hydrogen donor are available are severely limited.
In cyanobacterial photosynthesis, the needed energy is provided by light. However, it is noteworthy that many of the molecules that replace water as the hydrogen donor in cyanobacterial photosynthesis can also combine with oxygen to provide the energy needed to synthesize organic compounds in chemosynthesis (Chap. 12).