8.5: Absolute Age Dating
- Page ID
- 36923
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Numerical (or Absolute) Dating
Numerical (also known as absolute) dating deals with assigning actual dates (in years before the present) to geological events. This contrasts with relative dating, which instead is concerned with determining the order of events in Earth’s past. The science of absolute age dating is known as geochronology and the fundamental method of this science is radiometric dating (also known as isotopic dating).
Scholars and naturalists, understandably, have long been interested in knowing the absolute age of the Earth, as well as other important geological events. In 1650, Archbishop James Ussher (1581-1686) famously used the genealogy of the Old Testament of the Bible (e.g., Genesis, Chapter 5), and the human lifespans recorded in it, to estimate the age of the Earth. He concluded that the Earth was young in age, having formed in 4004 B.C., or about 6,000 years ago.

In the 1800s, practitioners of the young science of geology applied the uniformitarian views of Hutton and Lyell (discussed elsewhere in this chapter) to try to determine the age of the Earth. For example, some geologists observed how long it took for a given amount of sediment (say, a centimeter of sand) to accumulate in a modern habitat, then applied this rate to the total known thickness of sedimentary rocks. When they did this, they estimated that the Earth is many millions of years old.
We now know that this estimate is far too young. In part, this estimate is so low because these early geologists did not recognize that unconformities, which represent missing units of time, often caused by erosion, are rampant in the rock record, as well as the fact that some metamorphic rocks were once sedimentary, and thus left out of their calculations. Still, this estimate was on the order of millions of years, rather than Ussher’s calculation of 6,000 years. Geologists were beginning to accept the views of Hutton that the Earth is unimaginably ancient.
What key discovery, then, allowed geologists to begin assigning absolute age dates to rocks and ultimately discover the age of the Earth? The answer is radioactivity.
Radiometric Dating
Hypotheses of absolute ages of rocks (as well as the events that they represent) are determined from rates of radioactive decay of some isotopes of elements that occur naturally in rocks.
Elements and Isotopes
In chemistry, an element is a particular kind of atom that is defined by the number of protons it has in its nucleus. The number of protons equals the element’s atomic number. Have a look at the periodic table of the elements below. Carbon’s (C) atomic number is 6 because it has six protons in its nucleus; gold’s (Au) atomic number is 79 because it has 79 protons in its nucleus.

Even though individual elements always have the same number of protons, the number of neutrons in their nuclei sometimes varies. These variations are called isotopes. Isotopes of individual elements are defined by their mass number, which is the number of protons plus the number of neutrons.
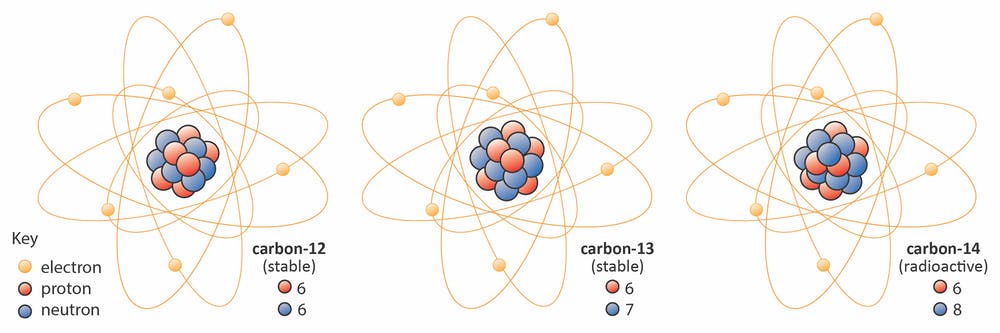
Consider, for example, the three different isotopes of Carbon each with 6 protons (which is what makes it Carbon):
- Carbon-12 (\(^{12}\)C): 6 protons, 6 neutrons
- Carbon-13 (\(^{13}\)C): 6 protons, 7 neutrons
- Carbon-14 (\(^{14}\)C): 6 protons, 8 neutrons
Radioactive Decay
Most isotopes are stable, meaning that they do not change, or decay, over time; they could remain the same stable isotope for eternity. Some isotopes, however, are unstable and undergo radical change through the process of radioactive decay. This involves the unstable isotopes shedding energy in the form of radiation and subatomic particles, causing their numbers of protons and neutrons to change. Once the number of protons change, the isotope is no longer the same element. The radioactive element will decay to form a different, stable element. The atomic nucleus that undergoes radioactive decay is known at the parent and the resulting stable element, the daughter product (or, decay product). Consider the example of the radioactive element \(\ce{^{238}U}\) (Uranium-238) which will radioactively decay to \(\ce{^{206}Pb}\) (Lead-206). This is not a one step process as indicated in the diagram below. We begin with \(\ce{^{238}U}\) in the upper left. As the decay process releases particles and energy, \(\ce{^{238}U}\) goes through a significant transformation, morphing into many other similarly radioactive elements prior to ending up as a stable isotope of lead, \(\ce{^{206}Pb}\). Some steps take only fractions of a second, others may take thousands of years, however, relative to the pace of “geologic time,” this process happens very quickly.

How do we apply the radioactive decay of unstable isotopes to the dating of rock? We are not really dating the rock, we are dating a mineral in the rock. Radioactive elements become part of the mineral chemistry during the crystallization process. The key to understanding how this works is to realize that the physical chemistry of a growing mineral crystal can be ‘picky’ or it can be ‘sloppy.’ When ‘picky’ minerals are forming, their growing mineral crystal lattice can be very “exclusive” about what atoms it will let into its crystal structure. It accepts a select few kinds of atoms, but turns away most every other kind. The opposite would be sloppy – where all sorts of different atoms are permitted into the developing lattice. Sloppy minerals aren’t going to be useful for geologic dating. We need picky minerals.
The ideal mineral for numerical dating accepts some atoms into its crystal that are radioactive parent isotopes. The parent will decay at a known rate, diminishing in number over time but causing their daughter (decay) products to accumulate over time in a one-for-one match. The ideal mineral loves bringing in the parent, but would never include the daughter in the developing crystal structure – like a “no children allowed” rule!
In some cases, the radioactive element in question is an isotope of an element essential to the mineral’s growth; one of its “standard ingredients” – for instance, \(\ce{^{40}K}\) getting incorporated into the ‘potassium spot’ in the potassium feldspar crystal (\({\bf{\ce{K}}}\ce{AlSi3O8}\)), where most of the atoms in that position are stable (not radioactive) isotopes of the same element, \(\ce{^{39}K}\) or \(\ce{^{41}K}\) in this case. \(\ce{^{40}K}\) is the radioactive parent. \(\ce{^{40}K}\) will decay to form \(\ce{^{40}Ar}\) as a stable daughter product. However, \(\ce{^{40}Ar}\), due to its size, will never be found in the original mineral. As radioactive decay of \(\ce{^{40}K} \rightarrow \ce{^{40}Ar}\) transpires over time, both elements remain trapped within the closed system of the formed mineral.
In other cases, we’re focused on elemental impurities in the mineral crystal – atoms you won’t find in the mineral’s common chemical formula. Specifically, picky minerals will (a) let certain radioactive atoms into their crystals, but (b) at the same time, they actively exclude the stable daughter atoms that result when the radioactive parents break down. The parent isotope “fits” into the crystal, by virtue of its valence state and its atomic size. Conversely, the daughter isotope doesn’t fit into the crystal, either because of its valence state or because of its atomic size, or both. An example of this occurs in the mineral zircon (\(\ce{ZrSiO4}\)). Stable atoms of Zr dominate in the crystal lattice however radioactive \(\ce{^{238}U}\) atoms can substitute for Zr. The daughter product of the decay of \(\ce{^{238}U}\) is \(\ce{^{206}Pb}\). Due to the atomic characteristics of Pb, there is no “happy place” for Pb in the crystal, so Pb atoms will never enter the zircon crystal lattice while it is forming. As radioactive decay of \(\ce{^{238}U} \rightarrow \ce{^{206}Pb}\) occurs over time, both elements remain trapped within the closed system of the formed mineral.
The parent-daughter decay pair found within the mineral crystal starts off with 0% daughter isotopes, and 100% radioactive parent isotopes. Based on the previous discussion, we can be confident that any daughter isotopes we find in the mineral crystal got there due to radioactive decay of a parent isotope some time after the crystal originally formed, and they were NOT there from the very beginning. They form in place, due to the spontaneous breakdown of their radioactive parent isotopes, and they are “trapped” in a mineral crystal in which they do not belong. A comprehensive count of all the parent isotopes and all the daughter isotopes in that mineral system will sum to 100%.
Minerals that meet these criteria are very few and far between. The most applicable ones are listed below on the Half-life Decay Constant Chart.
Half-life Decay Constant Chart
Table \(\PageIndex{1}\): Half-Life Decay Constant Chart
| Mineral | Radioactive parent that is “allowed” in the crystal | Daughter isotope that results | Half-life decay constant of the parent isotope | Useful age range in geologic dating | Useful in Igneous rocks | Useful in Sedimentary rocks | Useful in Metamorphic rocks |
|---|---|---|---|---|---|---|---|
| Zircon (also Monazite) | \(\ce{^{238}U}\) | \(\ce{^{206}Pb}\) | 4.5 billion years | More than 100 Ma | \(\checkmark\) | \(\checkmark\)* | \(\checkmark\)** |
| \(\ce{^{235}U}\) | \(\ce{^{207}Pb}\) | 703.8 million years | More than 100 Ma | \(\checkmark\) | \(\checkmark\)* | \(\checkmark\)** | |
| \(\ce{^{232}Th}\) | \(\ce{^{208}Pb}\) | 14 billion years – longer than the age of the Universe! | More than 200 Ma | \(\checkmark\) | \(\checkmark\)* | \(\checkmark\)** | |
| Muscovite, biotite, hornblende, potassium feldspar | \(\ce{^{40}K}\) | \(\ce{^{40}Ar}\) | 1.25 billion years | More than 100,000 years | \(\checkmark\) | \(\checkmark\) | |
| Muscovite, biotite, potassium feldspar | \(\ce{^{87}Rb}\) | \(\ce{^{87}Sr}\) | 48.8 billion years ~4 times older than the age of the Universe! | More than 100 Ma | \(\checkmark\) | \(\checkmark\)? |
*as detrital grains within a sedimentary deposit: gives a maximum age for the deposit.
**as overgrowths on original grains – must be distinguished from the mineral’s “core” during the dating process or as reset ages, if the rock has undergone very hot metamorphism (>800 \(^{\circ}\)C).
Did you notice how few minerals there are? Of the thousands of minerals known, only a handful are common enough in rocks and ‘picky’ enough in their chemistry to be useful for isotopic dating. Our toolbox has only about a dozen tools in it. We can’t numerically date quartz. We can’t numerically date calcite. Same for olivine and pyroxene and most of the important rock forming minerals.
Two pieces of information are needed to calculate how old a given mineral is: the ratio of parent to daughter isotopes, and the rate of decay, or half-life, of the radioactive parent.
- The ratio of parent to daughter atoms is measured by using a mass spectrometer which is a very sensitive, high precision instrument that detects and separates atoms based on their mass.
- The rate at which a particular parent isotope decays into its daughter product is constant. This rate is determined in a laboratory setting. A half-life is the amount of time needed for half of the parent atoms in a sample to decay into daughter products. This is illustrated in the chart below.
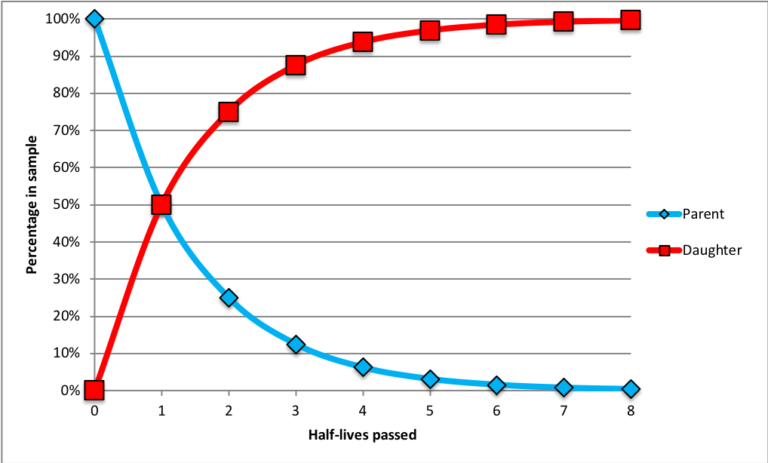
At the start time (zero half-lives passed), the sample consists of 100% parent atoms (blue diamonds); there are no daughter products (red squares) because no time has passed. After the passage of one half-life, 50% of the parent atoms have become daughter products. After two half-lives, 75% of the original parent atoms have been transformed into daughter products (thus, only 25% of the original parent atoms remain). After three half-lives, only 12.5% of the original parent atoms remain. As more half-lives pass, the number of parent atoms remaining approaches zero.
The ratio of parent atoms relative to daughter products in a sample will determine how many half-lives have passed since a mineral grain first formed. Consider the example shown below.

The left-most box in Figure \(\PageIndex{6}\) represents an initial state, with parent atoms distributed throughout molten rock (magma). As the magma cools, grains of different minerals begin to crystalize. Some of these minerals (represented above as gray hexagons) incorporate the radioactive parent atoms (blue diamonds) into their crystalline structures; this marks the initiation of the “half-life clock” (i.e., the start time, or time zero). After one half-life has passed, half (50%, or four) of the parent atoms in each mineral grain have been transformed into their daughter products (red squares). After two half-lives have passed, 75% (six) of the original parent atoms in each grain have been transformed into daughter products. How many parent atoms would remain if three half-lives passed? (Answer: only one as half of the two blue dots in each of the minerals would convert to daughter; so there would be one blue dot per crystal)
Calculating Radiometric Dates
The next step in radiometric dating involves converting the number of half-lives that have passed into a numerical (i.e., absolute) age. This is done by multiplying the number of half-lives that have passed by the half-life decay constant of the parent isotope (see the table above for decay constants).
Let’s step through a simple problem to determine the age of the sample of granite below.
Granite contains the mineral potassium feldspar which can be dated using the radioactive decay pair of \(\ce{^{40}K} \rightarrow \ce{^{40}Ar}\). We send our sample of potassium feldspar off to the lab to be analyzed using the latest mass spectrometry methods to determine the ratio of parent isotope of \(\ce{^{40}K}\) to daughter isotope of \(\ce{^{40}Ar}\). Analysis reveals that 75% of our parent \(\ce{^{40}K}\) remains and 25% of our daughter is present. We can use the half life chart (above) and the graph below to determine the age of the granite. The chart tells us that the half life of \(\ce{^{40}K}\) is 1.25 billion years. We read the graph as follows:

We can use either the percentage of the parent or the daughter; we will end up with the same answer. Choosing the daughter percentage of 25%, we find that along the left, vertical axis and then determine the intersection with the decay curve line for the daughter product (A). We drop down vertically from that point to the horizontal axis of half lives (B). The intersection of line B with the horizontal axis indicates that approximately 0.4 of one half life has passed since this sample originally formed. To determine the age of the sample in years, we simply multiply the half life decay constant for \(\ce{^{40}K} \rightarrow \ce{^{40}Ar}\) (1.25 By) by the number of half-lives passed (0.4):
\[1.25 \text{ By X } 0.4 = 500 \text{ My}\nonumber\]
To summarize, to determine the age of a sample you must:
1. Determine the percent of Parent/Daughter isotopes that exist within the sample.
2. Determine the number of half-lives that have passed by using the percent parent (or daughter) isotope and the Radioactive Decay Curve diagram.
3. Determine the age of the sample by multiplying the number of half-lives by the decay rate constant for the parent/daughter isotope pair found on the Half-life Decay Constant chart.
A Word About Carbon-14 (\(\ce{^{14}C}\)) Dating
Almost everyone has heard of Carbon-14 dating. \(\ce{^{14}C}\) does not appear on our Half-life Decay Constant chart above because it is not a useful isotope for dating rocks. \(\ce{^{14}C}\) has a very short half-life, geologically speaking. Its half-life is 5,730 years; compare that with the half-life data on the chart. After 5-6 half-lives (approximately 50,000 years), the amount of \(\ce{^{14}C}\) remaining is a sample becomes negligible and not useful for mineral/rock dating purposes. Carbon commonly exists in rock in the form of organic carbon and carbonate fossils. However the formation of lithified rock is in the order of millions of years therefore, any \(\ce{^{14}C}\) that originally existed in the sample has long since decayed to virtually nothing. As a result, \(\ce{^{14}C}\)’s main use is determining archeological dates of wood, charcoal, seeds, pollen, pottery, paper, natural fabric and more recent bone and shell material.
Mineral Ages vs. Rock Ages
How does the mineral’s numerical date relate to the rock’s numerical date? What considerations must the historical geologist bear in mind as they make the transfer from the one to the other? It depends on the rock, and it depends on the mineral.
The simplest case is with igneous rocks, wherein all the minerals form at the time molten rock (magma or lava) crystallizes into solid rock. So in igneous rocks, all the minerals are the same age as the rock. A zircon crystal (igneous mineral) date from a granite (igneous rock), therefore, may be taken as representative of the age of the granite. Exceptions would include xenoliths (pieces of older rock incorporated into igneous rocks) and xenocrysts (crystals from older rocks incorporated into igneous rocks).
In a clastic sedimentary rock, however, the situation is more complicated. Rocks like sandstone are made of sedimentary grains: small pieces of other rocks. Those other rocks must be older, in order to slough off pieces of themselves; pieces that can later be deposited with other small rock chunks and make a deposit of sand, sand that could then be lithified to make a sandstone. The minerals that make up a clastic sedimentary rock don’t form in place: they originated elsewhere, some time before. So a zircon crystal in a sedimentary rock does not represent a maximum age for the sedimentary deposit: its age is the age of the igneous rock in which it formed, so the sedimentary deposit which contains a bit of rock weathered out of that original igneous rock must be younger than it. If the zircon is 2 billion years old, then the sedimentary deposit containing it must be younger than 2 billion years old. But it could be 1 billion years old, or 100 million, or 100 years old. It could be 20 minutes old, and still contain a 2-billion-year-old zircon.

These are called “detrital zircons” to distinguish our understanding of how they got into the sedimentary rock – they are survivors from older weathered-away igneous source rocks. They are part of the leftover scraps – the detritus, or waste – from the breakdown of older rocks. Detrital zircon have been very useful in tracking down the Earth’s oldest rocks. At the beginning of this chapter, you learned that the Earth is 4.56 billion years old. As it turns out, the oldest dated mineral–a grain of zircon from the Jack Hills of Western Australia–is 4.404 billion years old. This zircon exists in metaconglomerate, a metamorphosed clastic sedimentary rock. Discovery of the age of this zircon tells us that it was derived from even older igneous rock which gives us a tremendous clue as to the composition of Earth’s early, Hadean age, crust.
In a chemical sedimentary rock, where crystals are forming at the time of deposition, it would be plausible that a mineral date could be used to date the depositional age, but the problem is that none of the crystals in chemical sedimentary rocks are “picky” enough about their isotopic inclusions for the technique to work. Calcite doesn’t grab radioactive isotopes while excluding their daughter isotopes, and neither does halite or gypsum. Tough luck: we just can’t date chemical sedimentary rocks using such a process. Next!
With metamorphic rocks, there’s another layer of complication still: the mineral age can be representative of the rock’s protolith, or the date of its metamorphism. It all depends on what mineral we’re talking about, and what the protolith was. First, let’s consider a metavolcanic rock – a metamorphic rock that started off as a volcanic (igneous) rock before it got metamorphosed. In this case, a zircon represents the age of the protolith (the age of the initial eruption), while the elevated temperature and pressure of metamorphism might encourage the growth of new metamorphic minerals. When a basalt is metamorphosed to make an amphibolite for instance, it grows new amphibole crystals. These amphiboles could be dated (using the K/Ar technique) in order to determine the age of metamorphism. Two minerals in one rock, each representative of a different process, happening at a distinct time. The careful geologist should pay attention to which mineral they are dating, and what its implications are for the history of that rock.
Next, consider a metasedimentary rock – a metamorphic rock with a sedimentary protolith. A metagraywacke is a metamorphosed graywacke: its protolith was clay-rich sandstone. In this case, a detrital zircon might well be a maximum age constraint of the original sedimentary deposit, as we just discussed a couple of paragraphs ago. Zircon has a very high closure temperature (link to resetting discussion below), so if the metamorphism was low- to medium-grade, the zircon’s original igneous date could survive heating. But the same level of metamorphism in the same metagraywacke could easily be sufficient that the clay all recrystallizes to make muscovite mica, then we can use the mica to get the age of metamorphism. The zircon would have been there before metamorphism, but the mica grew as a consequence of metamorphism.
An example of this can be found in the Piedmont metamorphic rocks of Washington, DC. There, detrital zircons in metagraywackes of the Sykesville Formation give U/Pb ages of ~1 Ga, but the same rocks also host metamorphic muscovites that grew at 460 Ma, according to the K/Ar dating technique. The conclusion is that the muddy sand was deposited sometime after 1 Ga, but before 460 Ma, since it was metamorphosed then. The two dating techniques, on two different minerals, help “bracket” the age of the original deposit of sediment.

One additional wrinkle with metamorphic rocks is that sometimes you can get several dates out of a single mineral crystal. This happens because an original crystal can add new layers when it gets metamorphosed. Again, we turn to zircon for an example: if a granite pluton was to be metamorphosed and make a gneiss, then original (igneous) zircons in the rock might have the opportunity to acquire new zirconium, silicon, and oxygen atoms through metamorphic recrystallization. These new atoms get arranged in the zircon’s distinctive crystal structure, accumulating on the outside of the old crystal. If you were to date this whole zircon crystal, you’re going to get a hybridized age – a mix of the original plutonic age and the age of the metamorphic overgrowth. That would be a disaster!
Fortunately, there is a solution: SHRIMP to the rescue! SHRIMP is an acronym that stands for Sensitive High-Resolution Ion MicroProbe. It’s a tool that allows the dating of different parts of a single zircon crystal. Although the crystal being examined may be the size of a grain of sand, the SHRIMP uses a thin laser beam to distinguish between the layers on the edge (youngest) versus the layers in the middle (oldest). Incredibly, this allows us to extract multiple dates from a single mineral grain: one date for the igneous crystallization, and another date for a later metamorphic event. There are even examples where 10 or more dates have been extracted from a single grain of zircon!
Metamorphism can complicate accurately dating the age of a particular mineral. Increased pressure and temperature during metamorphism plus the addition/liberation of fluids and gases can result in ions becoming mobile in the metamorphic environment. We can return to a previous discussion on calculating radiometric dates to use as an example. Potassium feldspar is one of the most common minerals in the crust; common in both igneous and metamorphic rocks. Potassium feldspar typically begins its existence as a mineral formed during the crystallization of magma. It is a common constituent in the igneous rock, granite. The radioactive isotope, \(\ce{^{40}K}\), can be included in the crystalline structure of potassium feldspar. Upon crystallization, the radioactive decay of \(\ce{^{40}K}\) begins producing \(\ce{^{40}Ar}\). Granite can be very accurately dated using this decay pair (\(\ce{^{40}K} / \ce{^{40}Ar}\)). However, most granite is exposed to metamorphic conditions because it typically forms in a cooling magma chamber, deep within the crust, in a tectonic environment that includes the compressive forces of plate collision. Granite, subjected to metamorphic conditions, does not change much in its mineralogy however the increased pressure and temperature conditions will realign minerals and allow trapped fluids and gasses to escape. The resulting metamorphic rock is a granitic gneiss. The realigned potassium feldspar minerals will remain in the granitic gneiss (see photo below) and still contain the decaying \(\ce{^{40}K}\). However, the daughter product, \(\ce{^{40}Ar}\), formed in the original granite, may have vacated the closed system during the metamorphic process thereby resetting the radiometric decay clock. Dating the potassium feldspar in the granitic gneiss would yield the date of metamorphism, not the original date potassium feldspar crystallization in the granite.

Here is a reminder of a few of the more complex concepts in this section.
- In order to date old objects, you must use an isotope with a large half-life, otherwise all of the parent decays to daughter product too quickly.
- Carbon-14 can not be used to date rocks like granite for two reasons. First, they are too old and Carbon-14's half-life is just under 6,000 years. This means it can't date materials past about 50,000 years old. Second, Carbon-14 is only found in living objects, and granite was never alive.
- Clastic sedimentary rocks can not be dated using isotopic age dating as they are made of pieces of other rocks. When you date a mineral in the clastic rock, it will produce the age of the rock it came from!
- Isotopic age dating of metamorphic rocks is possible but complicated. Since the "clock" is reset when the minerals get to a certain temperature during metamorphism and then decay starts once the temperature is lowered to a certain amount, problems can arise. Sometimes the "clock" is not completely reset and the rock has an age that is too old, reflecting the age of the protolith.
- daughter product – the stable atom that forms as a result of the decay of a radioactive parent isotope
- half-life – the amount of time it takes for half of the atoms in a sample of a radioactive isotope to decay into daughter products
- isotopes – atoms of the same element that have the same number of protons but different numbers of neutrons, giving them different atomic masses
- mass number – the total number of protons and neutrons in the nucleus of an atom
- numerical dating - the type of geologic dating in which you assign a number age to a geologic event
- parent – the original radioactive isotope that undergoes decay to form a daughter product
- radioactive decay – the natural process by which an unstable atomic nucleus loses energy by emitting particles or radiation, transforming into a more stable form (often producing a daughter isotope)


