17.1: Weathering
- Page ID
- 16632
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Slopes
A slope is an inclined ground surface. Most slopes are mantled with unconsolidated regolith, the product of weathering. Regolith serves as parent material for soil above and grades downward into unaltered bedrock below. Loose regolith serves as a source of colluvium, sediment that has been eroded, transported and deposited down slope. In order to do so, the erosional forces must overcome the forces of resistance: friction, inertia (the resistance to movement), and particle cohesion.
The land surface constantly responds to endogenic and exogenic forces that shape the Earth. Being an open system, a slope seeks a state of balance between the forces of resistance and those of change. The surface attains a state of dynamic equilibrium, a condition where the system constantly adjusts to processes that raise the surface (e.g. tectonic uplift) and those that wear it down (e.g. erosion by water). The system fluctuates around a stable average state unless the driving forces of change exceed a geomorphic threshold, such as a massive earthquake, tsunami, volcanism. Once this occurs, a period of system adjustment occurs, finally ending in a new stable state.
Weathering
Weathering is the breakdown and decomposition of earth material, namely rocks. Weathering is an important mechanism to destabilize surface materials for their eventual removal by erosive processes. Weathering of rock-forming minerals can create new products from pre-existing rocks. The physical disintegration of rocks affects soil development and texture. Weathering releases chemical compounds that become available for biological processes. The weathering of carbonate minerals releases carbon to the atmosphere which impacts atmospheric chemistry and temperature. And the list goes on. Weathering, needless to say, is an important environmental process that bridges all elements of our physical environment and sustains the notion of a changing Earth.

Weathering occurs in two ways. Physical weathering, also called mechanical weathering, involves the disintegration of rock materials. Physical weathering incurs no change in the chemistry of the material being altered. Instead, it simply breaks large pieces into smaller ones. Chemical weathering involves the decomposition of rocks and sediment. In this case, a chemical change occurs and a new product is created from the material that has undergone weathering. Weathering processes are determined by the climate and vegetation of a place. Dry locations tend to be dominated by physical weathering and moist places by chemical weathering.
Physical weathering
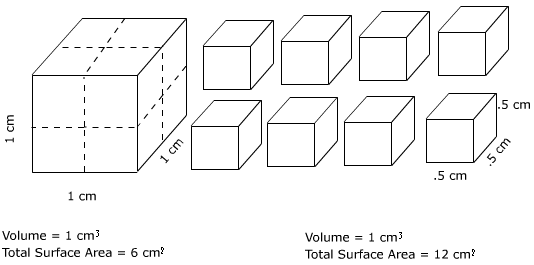
The result of physical weathering is to simply make smaller pieces out of larger ones. In so doing, physical weathering makes it easier for surface materials to chemically decompose and be eroded. When a large block of material is broken into smaller pieces additional surface area for chemical weathering to act is exposed. Examine Figure \(\PageIndex{2}\) below. On the left is a block whose length, width and height is equal to one centimeter. This means that the volume of the block is equal to 1 cubic centimeter and the total surface area is equal to 6 square centimeters. If we split the block in length and width wise (dashed lines) we create eight smaller pieces, each with a height, width, and depth of .5 centimeters. Though the volume of all eight pieces taken together is still 1 cubic centimeter, the total amount of surface area is greatly increased to 12 cm2.
Physical weathering processes
There are a number of physical weathering processes that break earth materials apart, a very common one is called root wedging. Plant roots work their way into rock crevices called joints. As they grow, roots create pressure on the sides of the crack enlarging it until the rock breaks apart. This is a common problem for home owners where trees are grow too close to a house. Tree roots can force their way into the foundation, breaking it apart, and let water seep into the owner's basement.

Frost wedging occurs when water freezes in rock fractures. As the water freezes it expands putting pressure on the sides of the crack, enlarging it until the rock breaks apart. Thermal expansion and contraction can weaken rock and cause it to disintegrate. In deserts, surface materials get exceedingly hot during the day and be exposed to cold temperatures at night. The expansion upon heating and contraction during cooling weakens rock breaking it apart. Alternate wetting and drying causes material to expand and contract, thus weakening rocks and inducing them to break as well. Regardless of process, the result is a mass of unconsolidated material.
Chemical weathering
The minerals in rocks formed beneath the surface are in equilibrium with the temperature and pressure conditions at time of their formation and thus are quite stable. However, many minerals are no longer in equilibrium with their environmental conditions when exposed at the surface and are susceptible to weathering. Chemical weathering results in the formation and retention of minerals in equilibrium with environmental conditions at the Earth's surface. The least stable minerals in igneous and metamorphic rocks are olivine and plagioclase, the most stable is quartz.
The interlocking and spacing of mineral grains controls the tendency towards weathering. Rocks with loosely interlocking mineral grains allow agents of chemical weathering to penetrate, thus speeding their decomposition. Limestone is primarily composed of calcite, a mineral that is quite soluble under surface conditions and easily dissolves in humid environments. In dry regions, the tight texture of limestone prevents it from disintegration and thus is a relatively resistant rock when found in deserts.
The process of chemical weathering tends to:
- increase bulk creating stress within rocks
- lower the density minerals
- decreased particle size resulting in increased surface area
- creates more mobile materials
- creates more stable minerals
Chemical weathering processes
Oxidation takes place when oxygen reacts with earth materials. Oxygen dissolved in water combines with atoms of metallic elements abundant in silicate minerals. Attacking metals in the soil, oxidation causes them to rust leaving the soil a brownish red to red color. When oxygen combines with iron, the reddish iron oxide hematite (Fe2O3) is formed:
\(4Fe^{+3}\,+\,3O_2\,\to\,2Fe_2O_3\)
Hydrolysis is an exchange reaction involving minerals and water. Free hydrogen (H+) and hydroxide (OH)- ions in water are able to replace mineral ions and drive them into solution. As a result, the mineral's atomic structure is changed into a new form. It is a process whereby silicate minerals like potassium feldspar are weathered and a clay mineral is formed.
\(2KAlSi_3O_8\,+\,2H^+\,+\,9H_2O\,\to\,Al_2Si_2O_5(OH)_4\,+\,4H_4SiO_4\,+\,2K^{2+}\)
Hydration involves the absorption of water like which occurs during the conversion of hematite to limonite:
\(2Fe_2O_3\,+\,3H_20\,\to\,2Fe_2O_3\,\cdot\,3H_20\)
Some geoscientists question whether hydration is a true chemical weathering process because the process is readily reversible and the new product is not chemically different from its precursor. Some would rather call hydration a physical weathering process.
Carbonic acid action involves combination of carbon dioxide and water. Though present in pure water, carbon dioxide dissolved in water provides ions that produces free hydrogen. Carbon dioxide in the atmosphere combines with rain water to form carbonic acid (H2CO3):
\(H_2O\,+\,CO_2\,\to\,H_2CO_3\)
Though weak, when carbonic acid is combined with a mineral like calcite (CaCO3) common to limestone, calcium and bicarbonate ions are released and carried off by groundwater.
\(CaCO_3\,+\,H_2CO_3\,\to\,Ca^{+2}\,+\,2HCO_{-3}\)
Rock resistance to weathering
Rocks react differently to weathering due to the differences in mineral content and structure. Some minerals are unstable under surface conditions and are readily soluble. Others are stable and resist the agents of weathering. Some rock-forming minerals are physically soft, being easily crushed and split while harder minerals are less easily broken apart. The arrangement and size of mineral grains control weathering processes. Water has a difficult time penetrating intricately locked and closely spaced mineral grains to promote weathering. Larger, loosely cemented minerals disintegrate and decompose more readily. Minerals in the form of poorly joined sheets readily break apart.
Granite is a coarse grained rock composed of quartz and feldspar. Both quartz and feldspar are hard minerals, but feldspar is less stable under surface conditions than quartz. The feldspar readily weathers to become clay in humid conditions. The feldspar will weather in dry climates as the somewhat porous granite allows moisture to penetrate. As the feldspar decomposes it weakens the bonds holding the rock together and it disintegrates
Karst Landscapes
The chemical weathering of carbonate-rich rocks creates a unique landscape abounding in caves, disappearing streams, and springs. Karst, a Yugoslavian term that comes from a narrow strip of limestone plateau noted for the assemblage of solution landforms. Karst develops in regions underlain by limestone and to a lesser extent dolomite. Chemical solution of the limestone, especially when fractured, wears away the bedrock leaving fissures and possibly undermining the surface. Some of the most spectacular cave complexes are found in such areas. Significant karst regions are found in Jamaica, the northern Yucatan, New South Wales, northern Puerto Rico, the Great Valley Region of Pennsylvania, Maryland, Virginia, and Tennessee, central Kentucky and central Florida in the United States

Generally four conditions are important for karst development. First, there must be limestone at or near the surface. Though karst is found in areas underlain by dolomite, it usually far less soluble than limestone. Second, the limestone must be dense, highly jointed, and thinly bedded. If the rock is too porous, water will be rapidly absorbed throughout the entire mass and not be concentrated along restricted flow lines. Third, the existence of entrenched valleys below uplands underlain by soluble and well jointed rocks ensures downward movement of groundwater, favorable for the development of karst. Finally, at least moderate precipitation must fall in the region. Few arid or semi-arid regions exhibit karst features, though some relic features may exist from a previous moist period in the past.
Karst Landforms
One of the most common features of areas underlain by limestone are sinkholes. Sinkholes form either from beneath the surface or from the surface down. Collapse sinkholes form when the limestone is dissolved away from below, removing the support for the surface and it collapses in. Collapse sinks can be particularly dangerous because one might not know that the support for the surface under their feet is being slowly eaten way. Sinkholes can also be created where water infiltrates into the surface widen the fissure into which it flows. These sinkholes are called dolines.
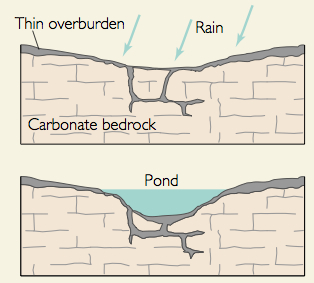
Sinkholes are commonly funnel-shaped and broadly open upward. They may be a few feet to more than 100 feet in depth, though usually ranging from 10 to 30 feet. Sinkhole diameter sizes range from a few square yards to several acres in area.
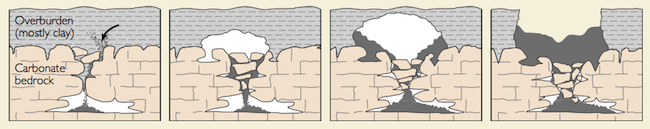
Streams flowing along the surface may enter a sinkhole as a "disappearing stream" and flow underground for some distance to reappear at the surface. A karst window forms when the roof above the underground stream collapses in. Cavern systems are carved by these underground streams over time.



