7.1: Getting Ready for Chapter 7
- Page ID
- 16043
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In Chapter 6 we learned how the atmosphere and oceans circulate air and water redistribute heat from regions of excess energy to those that are deficient. The energy that drives atmospheric circulation is largely that provided by the Sun. The unequal heating of the Earth creates temperature and pressure gradients that result in large scale circulation of the atmosphere. To a great extent, the major ocean currents are controlled by the circulation of the atmosphere. Atmospheric and oceanic circulation are the means of transport that enables the hydrologic cycle to function. As water is evaporated from the oceans or land, the circulation of the atmosphere moves it to another location where it can condense and precipitate out.
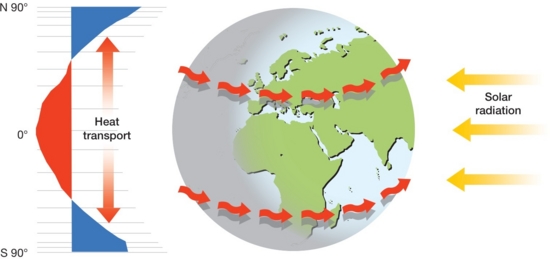
(Courtesy UNEP, Radiation. (2009). In UNEP/GRID-Arendal Maps and Graphics Library. Retrieved 19:17, December 21, 2009 from maps.grida.no/go/graphic/radiation.)
In Chapter 7, Atmospheric Moisture, we'll examine the processes that cause evaporation, condensation and the formation of precipitation. Using these new insights, we'll be able to explain the geographical distribution of atmospheric moisture. A look at how precipitation patterns may change as a result of human-induced climate change will conclude the chapter.
What you should already know ...
Understanding the processes that cause phase changes of water and control the distribution of pressure depends on knowledge gained in Chapter 2, Chapter 3, Chapter 4, and Chapter 6. In particular, you should have a handle on the basic terminology of heat and energy, how the radiation and energy balance relates to the water vapor content of the air, and how pressure systems influence air flow.
Use the quiz below to assess your understanding of a few key topics from these chapters.
- The heat used in the phase change of water is
- ground heat.
- latent heat.
- sensible heat.
- potential phase change heat.
- The average rate of decrease in temperature with an increase in elevation is
- the normal lapse rate of temperature.
- the environmental lapse rate of temperature.
- the latitudinal temperature gradient.
- none of the above.
- The greatest amount of latent heat transfer into the air for a year
- occurs over the midlatitude deserts.
- occurs near the Arctic ocean.
- occurs off the west coast of midlatitude continents.
- tropical oceans.
- The amount of energy required to raise the temperature of one gram of substance through 1oC is
- latent heat.
- potential heat.
- specific heat.
- sensible heat.
- A line that connects points of equal precipitation is called an
- isotach.
- isohyet.
- isobar.
- isobath.
- A cyclone exhibits
- converging surface air and subsidence.
- converging surface air and uplift.
- diverging surface air and uplift.
- diverging surface air and subsidence.
- The amount of heat required to raise the temperature of one gram of water through 1oC is called
- latent heat.
- specific heat.
- a calorie.
- a joule.
- Net radiation for the year is greatest
- over tropical oceans.
- off the west coast of the United States.
- over the Arctic ocean.
- in the Sahara desert.
- A change in air temperature without the physical exchange of heat is called
- a diabatic temperature change.
- an adiabatic temperature change.
- an environmental lapse rate.
- none of the above.
- A negative latent heat transfer
- means heat is removed from water molecules.
- means heat is added to water molecules.
- means the air temperature is high.
- none of the above.
- Answer
-
- B
- A
- D
- C
- B
- B
- C
- A
- B
- A
About your score ....
If you scored 80% or above, Great! ... start reading the chapter.
If you scored 70% to 80% you should consider reviewing the previous material.
If you scored less than 70% you should consider reviewing the previous material and seeking help from your instructor.


