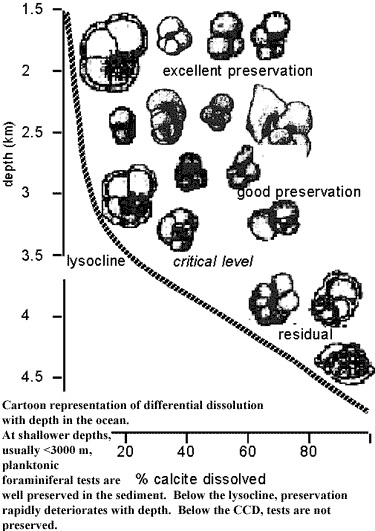
3.3: The Lysocline and the CCD
- Page ID
- 774
The lysocline and carbonate compensation depth (CCD) are two phenomena that affect the stability of calcite and aragonite in the deep ocean. The lysocline is the depth at which the rate of dissolution of calcite begins to increase dramatically. The water above the lysocline is supersaturated in calcite structures (\(\ce{CaCO3}\)), but as depth and pressure increase and temperature decreases, the solubility of calcite increases. This continues until the lysocline is reached. The lysocline is the point where there is a dramatic decrease (up to 90%) in the amount of calcite structures present, but below this exists the CCD. At the CCD the rate of supply of calcite equals the rate of dissolution, and no more calcite is deposited below this depth. In the Pacific, this depth is about 4,5000 below the surface; in the Atlantic, it is about 6,000 m deep. This dramatic variation is due to differences in ocean chemistry. The Pacific has a lower pH and is colder than the Atlantic, so its lysocline and CCD are higher in the water column because the solubility of calcite increases in these conditions.
This is why ocean acidification is such a major issue in modern oceanography. Because of our constant burning of our fossil fuels following the industrial revolution, we have dramatically increased the amount of \(\ce{CO2}\) in our atmosphere and essentially tilted the carbon cycle. This tilting of the carbon cycle has thrown off the equilibrium between the atmosphere and the ocean. By increasing the amount of \(\ce{CO2}\) in the atmosphere we have also increased the amount of \(\ce{CO2}\) in the ocean. By increasing the \(\ce{CO2}\) in the ocean we are increasing the amount of \(\ce{H^{+}}\) ions present.