3.1: The Essence of the Greenhouse Effect
- Page ID
- 11082
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Why does the absorption and emission of infrared radiation by the atmosphere warm the planet? This concept is actually quite easy to understand, though it is often explained poorly or even wrongly. When the greenhouse gases (and clouds, which also act as greenhouse agents) absorb infrared radiation, most of which comes from the surface and lower layers of the atmosphere, they must reemit radiation, otherwise the temperature of the atmosphere would increase indefinitely. This re-emission occurs in all directions, so that half the radiation is emitted broadly downward and half broadly upward. The downward part is absorbed by the earth’s surface or lower portions of the atmosphere. Thus, in effect, the earth’s surface receives radiant energy from two sources: the sun, and the backradiation from the greenhouse gases and clouds in the atmosphere, as illustrated in Figure \(\PageIndex{1}\). Now here is something surprising: on average the earth’s surface receives almost twice as much radiation from the atmosphere as it does directly from the sun, mostly because the atmosphere radiates 24/7, while the sun shines only part of the time. This is how powerful the greenhouse effect is.
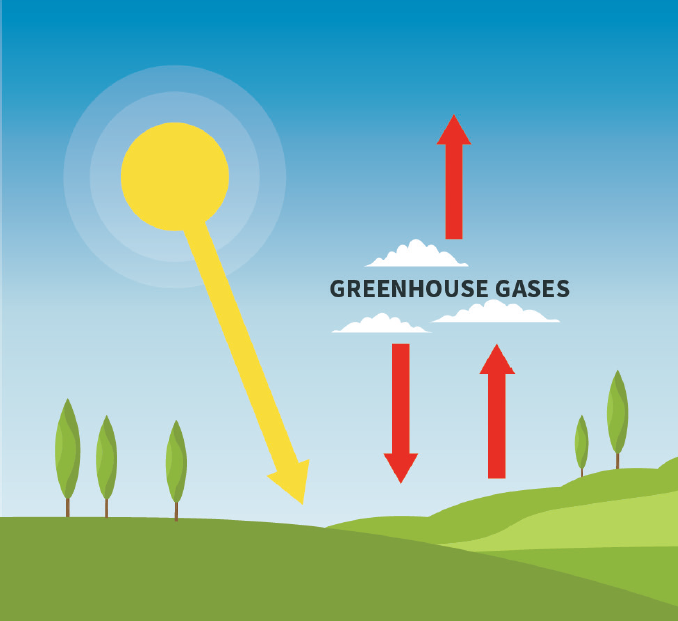
The surface must get warm enough to lose enough heat to balance both sunlight and back-radiation from the atmosphere and clouds within it. That is the greenhouse effect.
It should be remarked here that none of the preceding is remotely controversial among scientists, not even those few who express skepticism about global warming.
But not all greenhouse gases are created equal. The most important such gas, because of its relatively high concentrations, is water vapor, which can vary from almost nothing to as much as 3% of a volume of air. Also, condensed water (cloud) strongly absorbs and re-emits radiation, and reflects sunlight as well. Next to water, carbon dioxide has the largest effect on surface temperature, followed by methane and nitrous oxide, and a handful of other gases whose concentrations are truly minute.
Water is constantly exchanged between the atmosphere and the earth’s surface through evaporation and precipitation. This process is so rapid that, on average, a molecule of water resides in the atmosphere for only about two weeks.
The concentration of water vapor has an upper limit that is determined by air temperature—warmer air can support larger concentrations of water vapor. This is one reason that moisture varies so wildly from place to place and time to time. Another is that rain and snow can remove water from the air, so that its concentration can and often does fall well below the limit imposed by air temperature. The ratio of the actual amount of moisture in the air to its upper limit is what we refer to as relative humidity. Although relative humidity varies greatly, we observe that its long-term average is fairly stable, so to a first approximation, the actual amount of water in the atmosphere changes in tandem with its upper limit, that is, with temperature.
If we were magically to double the water vapor content of the atmosphere without changing its temperature, in roughly two weeks the excess water would be back where it belongs, in oceans, rivers, lakes, and groundwater. This would not be long enough to have much effect on climate. Temperature is the main determinant of the amount of water vapor in the atmosphere.
So, if the temperature rises, the amount of water vapor rises with it. But since water vapor is a greenhouse gas, rising water vapor leads to more back-radiation to the surface, which causes yet higher temperatures. We refer to this process as a positive feedback. Water vapor is thought to be the most important positive feedback in the climate system.
At the opposite extreme in terms of atmospheric lifetime is carbon dioxide. It is naturally emitted by volcanoes and absorbed by biological and physical processes that eventually incorporate the carbon into carbonate rocks like limestone. On geologic time scales, these rocks are subducted into the earth’s mantle at convergent boundaries of tectonic plates, and the carbon is eventually released back into the atmosphere as carbon dioxide through volcanoes or when the rock is once again exposed to air and weathered. This cycle takes many tens to hundreds of millions of years. But CO2 is exchanged between the atmosphere, ocean, and land plants on much shorter time scales. The bottom line is that if we were to instantly increase the concentration of CO2 in the atmosphere, roughly half of it would be absorbed back into plants and the upper ocean after only 100 years or so, but the other half might take many thousands of years to be removed from the air. For this reason, long-lived greenhouse gases like carbon dioxide have an important influence on climate.

