12.2: Non Renewable Energy Sources
- Page ID
- 11798
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Liquid Fossil Fuel: Petroleum
Thirty seven percent of the world’s energy consumption and 43 percent of the United States energy consumption comes from oil. Most of the oil production is in the Gulf region. Scientists and policy-makers often discuss the question of when the world will reach peak oil production, and there are a lot of variables in that equation, but it is generally thought that peak oil will be reached by the middle of the 21st Century. Currently world reserves are 1.3 trillion barrels, or 45 years left at current level of production, but we may reduce production as supplies run low.
Environmental Impacts of Oil Extraction and Refining
Oil is usually found one to two miles (1.6 – 3.2 km) below the surface. Oil refineries separate the mix of crude oil into the different types for gas, diesel fuel, tar, and asphalt. To find and extract oil workers must drill deep below ocean floor. As the United States tries to extract more oil from its own resources, we are drilling even deeper into the earth and increasing the environmental risks.
The largest United States oil spill to date began in April 2010 when an explosion occurred on Deepwater Horizon Oil Rig killing 11 employees and spilling nearly 200 million gallons of oil before the resulting leak could be stopped. Wildlife, ecosystems, and people’s livelihood were adversely affected. A lot of money and huge amounts of energy and waste were expended on immediate clean-up efforts. The long-term impacts are still not known. The National Commission on the Deepwater Horizon Oil Spill and Offshore Drilling was set up to study what went wrong.
Once oil is found and extracted it must be refined. Oil refining is one of top sources of air pollution in the United States for volatile organic hydrocarbons and toxic emissions, and the single largest source of carcinogenic benzene. When petroleum is burned as gasoline or diesel, or to make electricity or to power boilers for heat, it produces a number of emissions that have a detrimental effect on the environment and human health:
- Carbon dioxide (\(\ce{CO2}\)) is a greenhouse gas and a source of climate change.
- Sulfur dioxide (\(\ce{SO2}\)) causes acid rain, which damages plants and animals that live in water, and it increases or causes respiratory illnesses and heart diseases, particularly in vulnerable populations like children and the elderly.
- Nitrous oxides (\(\ce{NOx}\)) and Volatile Organic Carbons (VOCs) contribute to ozone at ground level, which is an irritatant and causes damage to the lungs.
- Particulate Matter (PM) produces hazy conditions in cities and scenic areas, and combines with ozone to contribute to asthma and chronic bronchitis, especially in children and the elderly. Very small, or “fine PM,” is also thought to penetrate the respiratory system more deeply and cause emphysema and lung cancer.
- Lead can have severe health impacts, especially for children.
- Air toxins are known or probable carcinogens.
There are other domestic sources of liquid fossil fuel that are being considered as conventional resources and are being depleted. These include soil sands/tar sands – deposits of moist sand and clay with 1-2 percent bitumen (thick and heavy petroleum rich in carbon and poor in hydrogen). These are removed by strip mining (see section above on coal). Another source is oil shale in United States west which is sedimentary rock filled with organic matter that can be processed to produce liquid petroleum. Also, mined by strip mines or subsurface mines, oil shale can be burned directly like coal or baked in the presence of hydrogen to extract liquid petroleum. However, the net energy values are low and they are expensive to extract and process. Both of these resources have severe environmental impacts due to strip mining, carbon dioxide, methane and other air pollutants similar to other fossil fuels.
Solid Fossil Fuel: Coal
Coal comes from organic matter that was compressed under high pressure to become a dense, solid carbon structure over thousands to millions of years. Due to its relatively low cost and abundance, coal is used to generate about half of the electricity consumed in the United States. Coal is the largest domestically produced source of energy. Figure Historic U.S. Coal Production shows how coal production has doubled in the United States over the last sixty year. Current world reserves are estimated at 826,000 million tonnes, with nearly 30 percent of that in the United States. It is a major fuel resource that the United States controls domestically.
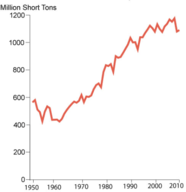 Figure \(\PageIndex{1}\): Historic U.S. Coal Production Graph shows U.S. Coal Production from 1950-2010. Source: U.S. Energy Information Administration
Figure \(\PageIndex{1}\): Historic U.S. Coal Production Graph shows U.S. Coal Production from 1950-2010. Source: U.S. Energy Information Administration
Coal is plentiful and inexpensive, when looking only at the market cost relative to the cost of other sources of electricity, but its extraction, transportation, and use produces a multitude of environmental impacts that the market cost does not truly represent. Coal emits sulfur dioxide, nitrogen oxide, and mercury, which have been linked to acid rain, smog, and health issues. Burning of coal emits higher amounts of carbon dioxide per unit of energy than the use of oil or natural gas. Coal accounted for 35 percent of the total United States emissions of carbon dioxide released into the Earth’s atmosphere in 2010. Ash generated from combustion contributes to water contamination. Some coal mining has a negative impact on ecosystems and water quality, and alters landscapes and scenic views. There are also significant health effects and risks to coal miners and those living in the vicinity of coal mines.
Traditional underground mining is risky to mine workers due to the risk of entrapment or death. Over the last 15 years, the U.S. Mine Safety and Health Administration has published the number of mine worker fatalities and it has varied from 18-48 per year.
Twenty-nine miners died on April 6, 2010 in an explosion at the Upper Big Branch coal mine in West Virginia, contributing to the uptick in deaths between 2009 and 2010. In other countries, with less safety regulations, accidents occur more frequently. In May 2011, for example, three people died and 11 were trapped in a coalmine in Mexico for several days. There is also risk of getting black lung disease (pneumoconiosis) This is a disease of the lungs caused by the inhalation of coal dust over a long period of time. It causes coughing and shortness of breath. If exposure is stopped the outcome is good. However, the complicated form may cause shortness of breath that gets increasingly worse.
Mountain Top Mining (MTM), while less hazardous to workers, has particularly detrimental effects on land resources. MTM is a surface mining practice involving the removal of mountaintops to expose coal seams, and disposing of the associated mining waste in adjacent valleys – "valley fills."
 Figure \(\PageIndex{2}\): Mountaintop Removal Coal Mining in Martin County, Kentucky Photograph shows mountaintop coal removal mining in Martin County, Kentucky. Source: Flashdark.
Figure \(\PageIndex{2}\): Mountaintop Removal Coal Mining in Martin County, Kentucky Photograph shows mountaintop coal removal mining in Martin County, Kentucky. Source: Flashdark.
The following are some examples of the impact of MTM:
- an increase of minerals in the water that negatively impact fish and macroinvertebrates, leading to less diverse and more pollutant-tolerant species
- streams are sometimes covered up by silt from mining
- the re-growth of trees and woody plants on regraded land may be slowed due to compacted soils
- affects the diversity of bird and amphibian species in the area since the ecosystem changes from wooded areas to other
- there may be social, economic and heritage issues created by the loss of wooded land that may have been important to traditions and economies of the area
Gaseous Fossil Fuel: Natural Gas
Natural gas meets 20 percent of world energy needs and 25 percent of United States needs. Natural gas is mainly composed of methane, the shortest hydrocarbon (\(\ce{CH4}\)), and is a very potent greenhouse gas. There are two types of natural gas. Biogenic gas is found at shallow depths and arises from anaerobic decay of organic matter by bacteria, like landfill gas. Thermogenic gas comes from the compression of organic matter and deep heat underground. They are found with petroleum in reservoir rocks and with coal deposits, and these fossil fuels are extracted together.
Methane is released into the atmosphere from coal mines, oil and gas wells, and natural gas storage tanks, pipelines, and processing plants. These leaks are the source of about 25 percent of total U.S. methane emissions, which translates to three percent of total U.S. greenhouse gas emissions. When natural gas is produced but cannot be captured and transported economically, it is "flared," or burned at well sites. This is considered to be safer and better than releasing methane into the atmosphere because \(\ce{CO2}\) is a less potent greenhouse gas than methane.
In the last few years a new reserve of natural gas has been identified - shale resources. The United States possesses 2,552 trillion cubic feet (Tcf) (72.27 trillion cubic meters) of potential natural gas resources, with shale resources accounting for 827 Tcf (23.42 tcm). As gas prices increased it has become more economical to extract the gas from shale. Figure U.S. Natural Gas Supply, 1990-2035 shows the past and forecasted U.S. natural gas production and the various sources. The current reserves are enough to last about 110 years at the 2009 rate of U.S. consumption (about 22.8 Tcf per year -645.7 bcm per year).
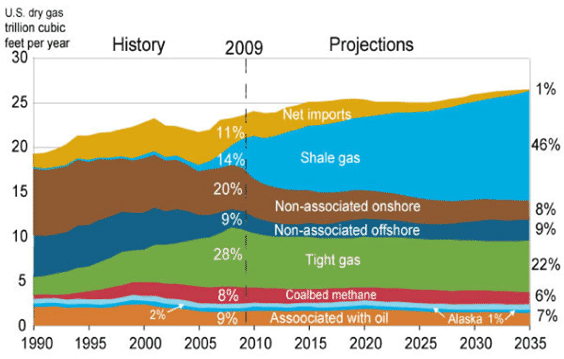 Figure \(\PageIndex{3}\): U.S. Natural Gas Supply, 1990-2035 Graph shows U.S. historic and projected natural gas production from various sources. Source: U.S. Energy Information Administration
Figure \(\PageIndex{3}\): U.S. Natural Gas Supply, 1990-2035 Graph shows U.S. historic and projected natural gas production from various sources. Source: U.S. Energy Information Administration
Natural gas is a preferred energy source when considering its environmental impacts. Specifically, when burned, much less carbon dioxide (\(\ce{CO2}\)), nitrogen oxides, and sulfur dioxide are omitted than from the combustion of coal or oil. It also does not produce ash or toxic emissions.
Environmental Impacts of Exploration, Drilling, and Production
Land resources are affected when geologists explore for natural gas deposits on land, as vehicles disturb vegetation and soils. Road clearing, pipeline and drill pad construction also affect natural habitats by clearing and digging. Natural gas production can also result in the production of large volumes of contaminated water. This water has to be properly handled, stored, and treated so that it does not pollute land and water supplies. Extraction of shale gas is more problematic than traditional sources due to a process nicknamed fracking or fracturing of wells, since it requires large amounts of water (see Figure below). The technique uses high-pressure fluids to fracture the normally hard shale deposits and release gas and oil trapped inside the rock. To promote the flow of gas out of the rock, small particles of solids are included in the fracturing liquids to lodge in the shale cracks and keep them open after the liquids are depressurized. The considerable use of water may affect the availability of water for other uses in some regions and this can affect aquatic habitats. If mismanaged, hydraulic fracturing fluid can be released by spills, leaks, or various other exposure pathways. The fluid contains potentially hazardous chemicals such as hydrochloric acid, glutaraldehyde, petroleum distillate, and ethylene glycol. The risks of fracking have been highlighted in popular culture in the documentary, Gasland (2010).
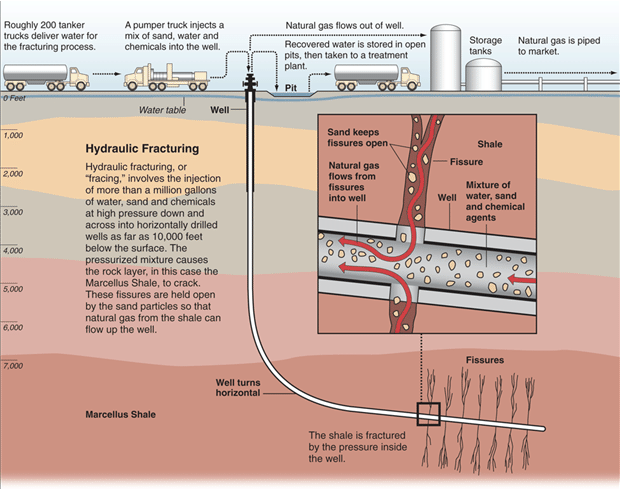 Figure \(\PageIndex{4}\): Graphic illustrates the process of hydraulic fracturing. Source: Al Granberg, ProPublica.
Figure \(\PageIndex{4}\): Graphic illustrates the process of hydraulic fracturing. Source: Al Granberg, ProPublica.
Fracking also produces large amounts of wastewater, which may contain dissolved chemicals from the hydraulic fluid and other contaminants that require treatment before disposal or reuse. Because of the quantities of water used and the complexities inherent in treating some of the wastewater components, treatment and disposal is an important and challenging issue.
The raw gas from a well may contain many other compounds besides the methane that is being sought, including hydrogen sulfide, a very toxic gas. Natural gas with high concentrations of hydrogen sulfide is usually flared which produces CO2, carbon monoxide, sulfur dioxide, nitrogen oxides, and many other compounds. Natural gas wells and pipelines often have engines to run equipment and compressors, which produce additional air pollutants and noise.
Contributions of Coal and Gas to Electricity Generation
At present the fossil fuels used for electricity generation are predominantly coal (45 percent) and gas (23 percent); petroleum accounts for approximately 1 percent. Coal electricity traces its origins to the early 20th Century, when it was the natural fuel for steam engines given its abundance, high energy density and low cost. Gas is a later addition to the fossil electricity mix, arriving in significant quantities after World War II and with its greatest growth since 1990. Of the two fuels, coal emits almost twice the carbon dioxide as gas for the same heat output, making it significantly greater contributor to global warming and climate change.
The Future of Gas and Coal
The future development of coal and gas depend on the degree of public and regulatory concern for carbon emissions, and the relative price and supply of the two fuels. Supplies of coal are abundant in the United States, and the transportation chain from mines to power plants is well established by long experience. The primary unknown factor is the degree of public and regulatory pressure that will be placed on carbon emissions. Strong regulatory pressure on carbon emissions would favor retirement of coal and addition of gas power plants. This trend is reinforced by the recent dramatic expansion of shale gas reserves in the United States due to advances in horizontal drilling and hydraulic fracturing of shale gas fields. Shale gas production has increased 48 percent annually in the years 2006 – 2010, with more increases expected. Greater United States production of shale gas will gradually reduce imports and could eventually make the United States a net exporter of natural gas.
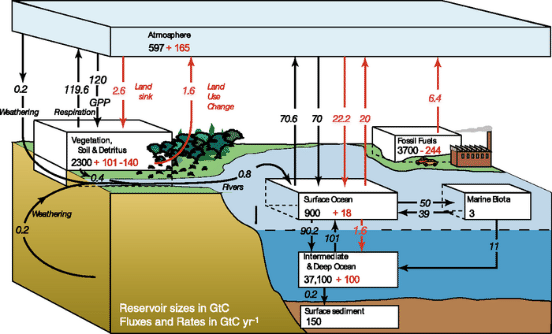 Figure \(\PageIndex{5}\): Global Carbon Cycle, 1990s The global carbon cycle for the 1990s, showing the main annual fluxes in GtC yr–1: pre-industrial ‘natural’ fluxes in black and ‘anthropogenic’ fluxes in red. Source: Climate Change 2007: The Physical Science Basis: Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, figure 7.3
Figure \(\PageIndex{5}\): Global Carbon Cycle, 1990s The global carbon cycle for the 1990s, showing the main annual fluxes in GtC yr–1: pre-industrial ‘natural’ fluxes in black and ‘anthropogenic’ fluxes in red. Source: Climate Change 2007: The Physical Science Basis: Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, figure 7.3
Beyond a trend from coal to gas for electricity generation, there is a need to deal with the carbon emissions from the fossil production of electricity. Figure above shows the size of these emissions compared to natural fluxes between ocean and atmosphere and from vegetation and land use. The anthropogenic fluxes are small by comparison, yet have a large effect on the concentration of carbon dioxide in the atmosphere. The reason is the step-wise dynamics of the carbon cycle. The ultimate storage repository for carbon emissions is the deep ocean, with abundant capacity to absorb the relatively small flux from fossil fuel combustion. Transfer to the deep ocean, however, occurs in three steps: first to the atmosphere, then to the shallow ocean, and finally to the deep ocean. The bottleneck is the slow transfer of carbon dioxide from the shallow ocean to the deep ocean, governed by the great ocean conveyor belt or thermohaline circulation illustrated in Figure below. The great ocean conveyor belt takes 400 – 1000 years to complete one cycle. While carbon dioxide waits to be transported to the deep ocean, it saturates the shallow ocean and "backs up" in the atmosphere causing global warming and threatening climate change. If carbon emissions are to be captured and stored (or "sequestered") they must be trapped for thousands of years while the atmosphere adjusts to past and future carbon emissions.
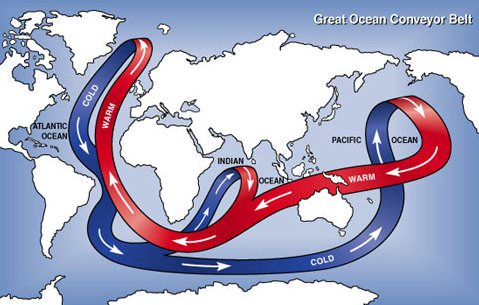 Figure \(\PageIndex{6}\): Great Ocean Conveyor Belt The great ocean conveyor belt (or thermohaline current) sends warm surface currents from the Pacific to Atlantic oceans and cold deep currents in the opposite direction. The conveyor belt is responsible for transporting dissolved carbon dioxide from the relatively small reservoir of the shallow ocean to much larger reservoir of the deep ocean. It takes 400 - 1000 years to complete one cycle. Source: Argonne National Laboratory
Figure \(\PageIndex{6}\): Great Ocean Conveyor Belt The great ocean conveyor belt (or thermohaline current) sends warm surface currents from the Pacific to Atlantic oceans and cold deep currents in the opposite direction. The conveyor belt is responsible for transporting dissolved carbon dioxide from the relatively small reservoir of the shallow ocean to much larger reservoir of the deep ocean. It takes 400 - 1000 years to complete one cycle. Source: Argonne National Laboratory
Sequestration of carbon dioxide in underground geologic formations is one process that, in principle, has the capacity to handle fossil fuel carbon emissions, chemical reaction of carbon dioxide to a stable solid form is another. For sequestration, there are fundamental challenges that must be understood and resolved before the process can be implemented on a wide scale.
The chemical reactions and migration routes through the porous rocks in which carbon dioxide is stored underground are largely unknown. Depending on the rock environment, stable solid compounds could form that would effectively remove the sequestered carbon dioxide from the environment. Alternatively, it could remain as carbon dioxide or transform to a mobile species and migrate long distances, finally finding an escape route to the atmosphere where it could resume its contribution to greenhouse warming or cause new environmental damage. The requirement on long term sequestration is severe: a leak rate of 1 percent means that all the carbon dioxide sequestered in the first year escapes in a century, a blink of the eye on the timescale of climate change.
Nuclear Power
Nuclear power plants produce no carbon dioxide and, therefore, are often considered an alternative fuel, when the main concern is climate change. Currently, world production is about 19.1 trillion KWh, with the United States producing and consuming about 22 percent of that. Nuclear power provides about nine percent of our total consumption for electricity (see Figure below).
However, there are environmental challenges with nuclear power. Mining and refining uranium ore and making reactor fuel demands a lot of energy. The plants themselves are made of metal and concrete which also requires energy to make. The main environmental challenge for nuclear power is the wastes including uranium mill tailings, spent (used) reactor fuel, and other radioactive wastes. These materials have long radioactive half-lives and thus remain a threat to human health for thousands of years. The U.S. Nuclear Regulatory Commission regulates the operation of nuclear power plants and the handling, transportation, storage, and disposal of radioactive materials to protect human health and the environment.
By volume, uranium mill tailings are the largest waste and they contain the radioactive element radium, which decays to produce radon, a radioactive gas. This waste is placed near the processing facility or mill where they come from, and are covered with a barrier of a material such as clay to prevent radon from escaping into the atmosphere and then a layer of soil, rocks, or other materials to prevent erosion of the sealing barrier.
High-level radioactive waste consists of used nuclear reactor fuel. This fuel is in a solid form consisting of small fuel pellets in long metal tubes and must be stored and handled with multiple containment, first cooled by water and later in special outdoor concrete or steel containers that are cooled by air. There is no long-term storage facility for this fuel in the United States.
There are many other regulatory precautions governing permitting, construction, operation, and decommissioning of nuclear power plants due to risks from an uncontrolled nuclear reaction. The potential for contamination of air, water and food is high should an uncontrolled reaction occur. Even when planning for worst-case scenarios, there are always risks of unexpected events. For example, the March 2011 earthquake and subsequent tsunami that hit Japan resulted in reactor meltdowns at the Fukushima Daiichi Nuclear Power Station causing massive damage to the surrounding area.
Note
Fukushima Daiichi Nuclear Power Station
- March 11, 2011: Magnitude 9.0 earthquake 231 miles northeast of Tokyo. Less than 1 hour later a 14m tsunami hit
- 50 power station employees worked around the clock to try to stabilize the situation
United States’ nuclear reactors have containment vessels that are designed to withstand extreme weather events and earthquakes. However, in the aftermath of the Japan incident, they are reviewing their facilities, policies, and procedures.
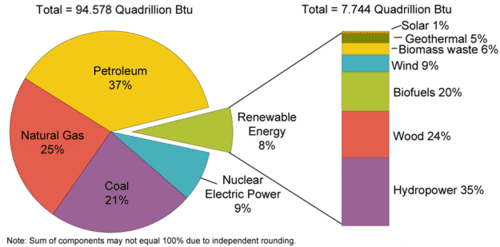 Figure \(\PageIndex{7}\): U.S. Energy Consumption by Energy Source, 2009 Renewable energy makes up 8% of U.S. energy consumption. Source: U.S. Energy Information Administration
Figure \(\PageIndex{7}\): U.S. Energy Consumption by Energy Source, 2009 Renewable energy makes up 8% of U.S. energy consumption. Source: U.S. Energy Information Administration
Debating Nuclear Energy
From a sustainability perspective, nuclear electricity presents an interesting dilemma. On the one hand, nuclear electricity produces no carbon emissions, a major sustainable advantage in a world facing human induced global warming and potential climate change. On the other hand, nuclear electricity produces spent fuel that must be stored out of the environment for tens or hundreds of thousands of years, it produces bomb-grade plutonium and uranium that could be diverted by terrorists or others to destroy cities and poison the environment, and it threatens the natural and built environment through accidental leaks of long lived radiation. Thoughtful scientists, policy makers and citizens must weigh the benefit of this source of carbon free electricity against the environmental risk of storing spent fuel for thousands or hundreds of thousands of years, the societal risk of nuclear proliferation, and the impact of accidental releases of radiation from operating reactors. There are very few examples of humans having the power to permanently change the dynamics of the earth. Global warming and climate change from carbon emissions is one example, and radiation from the explosion of a sufficient number of nuclear weapons is another. Nuclear electricity touches both of these opportunities, on the positive side for reducing carbon emissions and on the negative side for the risk of nuclear proliferation.
Nuclear electricity came on the energy scene remarkably quickly. Following the development of nuclear technology at the end of World War II for military ends, nuclear energy quickly acquired a new peacetime path for inexpensive production of electricity. Eleven years after the end of World War II, in 1956, a very short time in energy terms, the first commercial nuclear reactor produced electricity at Calder Hall in Sellafield, England. The number of nuclear reactors grew steadily to more than 400 by 1990, four years after the Chernobyl disaster in 1986 and eleven years following Three Mile Island in 1979. Since 1990, the number of operating reactors has remained approximately flat, with new construction balancing decommissioning, due to public and government reluctance to proceed with nuclear electricity expansion plans. Figure Growth of Fuels Used to Produce Electricity in the United States and Figure Nuclear Share of United States Electricity Generation show the development and status of nuclear power in the United States, a reflection of its worldwide growth.
The outcome of this debate will determine whether the world experiences a nuclear renaissance that has been in the making for several years. The global discussion has been strongly impacted by the unlikely nuclear accident in Fukushima, Japan in March 2011. The Fukushima nuclear disaster was caused by an earthquake and tsunami that disabled the cooling system for a nuclear energy complex consisting of operating nuclear reactors and storage pools for underwater storage of spent nuclear fuel ultimately causing a partial meltdown of some of the reactor cores and release of significant radiation. This event, 25 years after Chernobyl, reminds us that safety and public confidence are especially important in nuclear energy; without them expansion of nuclear energy will not happen.
There are two basic routes for handling the spent fuel of nuclear reactors: once through and reprocessing. Once through stores spent fuel following a single pass through the reactor, first in pools at the reactor site while it cools radioactively and thermally, then in a long-term geologic storage site, where it must remain for hundreds of thousands of years. Reprocessing separates the useable fraction of spent fuel and recycles it through the reactor, using a greater fraction of its energy content for electricity production, and sends the remaining high-level waste to permanent geologic storage. The primary motivation for recycling is greater use of fuel resources, extracting ~ 25 percent more energy than the once through cycle. A secondary motivation for recycling is a significant reduction of the permanent geologic storage space (by a factor of ~ 5 or more) and time (from hundreds of thousands of years to thousands of years). While these advantages seem natural and appealing from a sustainability perspective, they are complicated by the risk of theft of nuclear material from the reprocessing cycle for use in illicit weapons production or other non-sustainable ends. At present, France, the United Kingdom, Russia, Japan and China engage in some form of reprocessing; the United States, Sweden and Finland do not reprocess.
The Global Dependence of Transportation on Oil
Liquid petroleum fuels and electricity are the two dominant energy carriers in the United States, oil accounting for 37 percent of primary energy and electricity for 38 percent. These two energy carriers account for a similar fraction of carbon emissions, 36 percent and 38 percent, respectively. Two thirds of oil consumption is devoted to transportation, providing fuel for cars, trucks, trains and airplanes. For the United States and most developed societies, transportation is woven into the fabric of our lives, a necessity as central to daily operations as food or shelter. The concentration of oil reserves in a few regions or the world (Figure Crude Oil Reserves) makes much of the world dependent on imported energy for transportation.
The rise in the price of oil in the last decade makes dependence on imported energy for transportation an economic as well as an energy issue. The United States, for example, now spends upwards of $350 billion annually on imported oil, a drain of economic resources that could be used to stimulate growth, create jobs, build infrastructure and promote social advances at home.
From a sustainability perspective, oil presents several challenges. First is the length of time over which the world's finite oil reserves can continue to supply rising demand. Second is the impact on global warming and climate change that carbon emissions from oil combustion will have, and third is the challenge of finding a sustainable replacement for oil for transportation. Although we know the general course of initial rise and ultimate fall that global oil production must take, we do not know with confidence the time scale over which it will play out.
The uncertainty of the timing of the peak in global oil production encourages us to find other issues and motivations for dealing with an inevitably unsustainable supply. A prime motivation is energy security, the threat that oil supplies could be interrupted by any of several events including weather, natural disaster, terrorism and geopolitics. Much of the world feels these threats are good reasons for concerted effort to find replacements for oil as our primary transportation fuel. A second motivation is the environmental damage and accumulation of greenhouse gases in the atmosphere due to transportation emissions. Unlike electricity generation, transportation emissions arise from millions of tiny sources, e.g. the tailpipes of cars and trucks and the exhaust of trains and airplanes.
The challenge of capturing and sequestering carbon dioxide from these distributed and moving sources is dramatically greater than from the large fixed sources of power plants. A more achievable objective may be replacing oil as a transportation fuel with biofuel that recycles naturally each year from tailpipes of cars to biofuel crops that do not compete with food crops. Other options include replacing liquid fuels with electricity produced domestically, or increasing the efficiency of vehicles by reducing their weight, regeneratively capturing braking energy, and improving engine efficiency. Each of these options has promise and each must overcome challenges.
Changes in the energy system are inevitably slow, because of the time needed to develop new technologies and the operational inertia of phasing out the infrastructure of an existing technology to make room for a successor. The transportation system exhibits this operational inertia, governed by the turnover time for the fleet of vehicles, about 15 years. Although that time scale is long compared to economic cycles, the profit horizon of corporations and the political horizon of elected officials, it is important to begin now to identify and develop sustainable alternatives to oil as a transportation fuel. The timescale from innovation of new approaches and materials to market deployment is typically 20 years or more, well matched to the operational inertia of the transportation system. The challenge is to initiate innovative research and development for alternative transportation systems and sustain it continuously until the alternatives are established.

