3.1: Front Matter - Minerals and Mineral Identification
- Page ID
- 14785
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Have You Used a Mineral Today?
While many people may initially say no, consider these questions: Have you brushed your teeth? Have you eaten anything that contained salt? Did you put on make-up this morning? Have you painted your fingernails or toenails? Used a cellphone or other electronic device? Used a form of transportation, car, bike, or public transit? A yes to any of these questions, means you have used at least one mineral today, and likely probably many more! Minerals are useful and common in everyday products, yet most people do not even realize it (Figure 3.1).
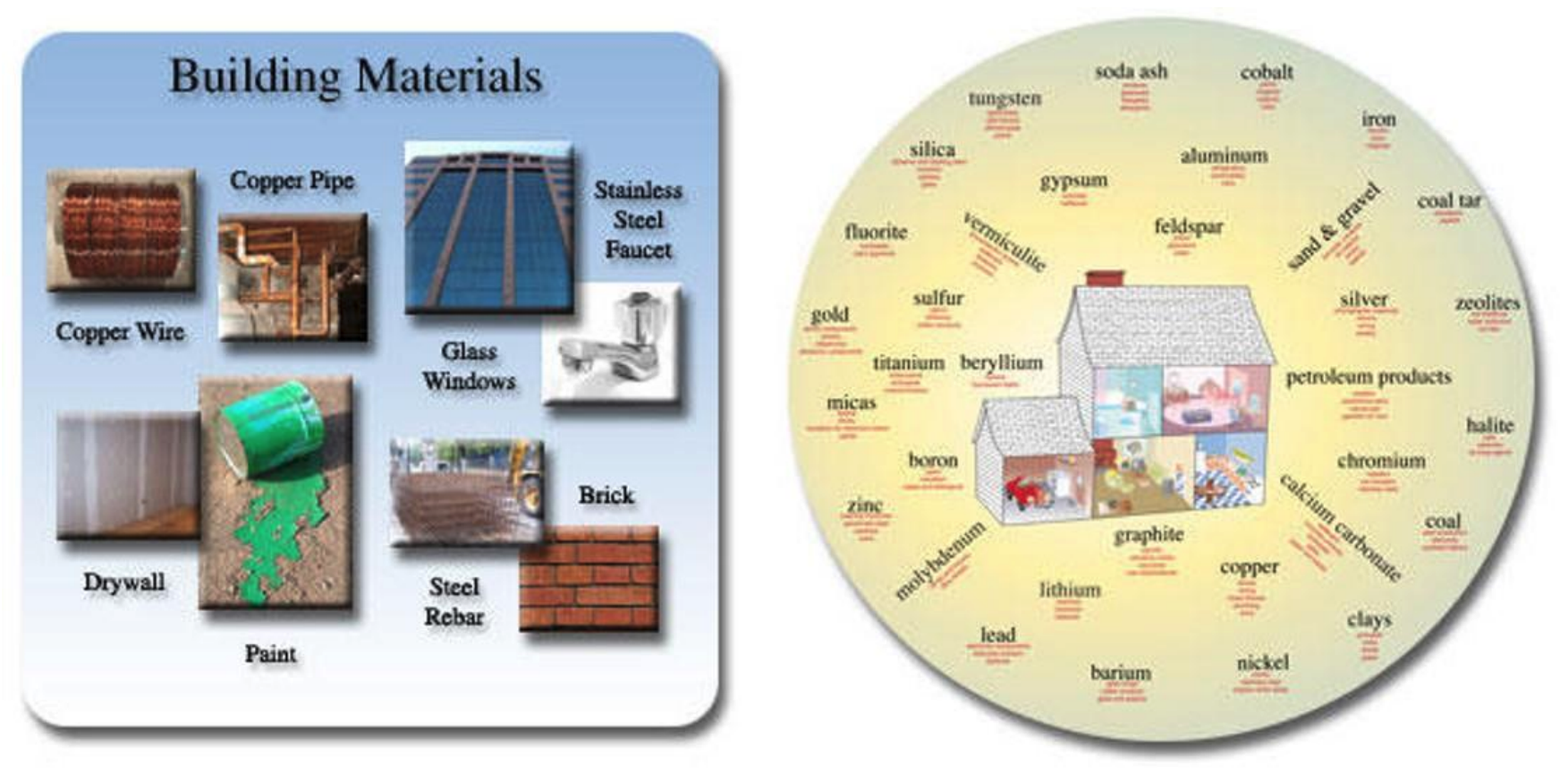
Figure 3:1: Common mined building materials, including minerals and other earth resources.
Minerals are not only important for their many uses, but also as the building blocks of rocks. In this lab, you will lay the foundation for many future labs in the course. Correct mineral identification is critical in geology, so work through this lab carefully. There are several thousand minerals, but we will focus on only the most common ones.
The geoscientists who study minerals are called mineralogists. Like many other geoscientists, working with other disciplines is common, with a heavy influence from both math and technology. Many are employed by universities where they teach and/or do research, and state and federal agencies, including geological surveys, like the California Geological Survey or United State Geological Survey (USGS). Additional career pathways are available in the private sector including in mining and natural resource extraction. Many of these career options require a college degree and postgraduate work (typically a PhD). If you are interested in mineralogy talk to your geology instructor for advice. We recommend completing as many math and science courses as possible (chemistry is incredibly important for mineralogy). Also, visit National Parks, CA State Parks, museums, gem & mineral shows, or join a local rock and mineral club. Typically, natural history museums will have wonderful displays of rocks, including those from your local region. Here in California, there are a number of large collections, including the San Diego Natural History Museum, Natural History Museum of Los Angeles County, Santa Barbara Museum of Natural History, and Kimball Natural History Museum. Many colleges and universities also have their own collections/museums.
What Is a Mineral?
A mineral is defined as any naturally occurring, inorganic solid with a definite chemical composition and a characteristic crystalline structure. Let’s break down this definition down.
-
Naturally occurring, means that anything synthetic (human-made), does not count as a mineral.
-
To be an inorganic solid, the mineral must not be composed of the complex carbon molecules that are characteristic of life and must be in the solid state, rather than vapor or liquid. This means that water, a liquid, is not a mineral, while water-ice, a solid, would be a mineral, provided it is out in nature and not synthetic.
-
A definite chemical composition refers to a definable chemical formula. Some minerals will have short chemical formulas, like Halite (NaCl or sodium chloride). For other minerals, like Olivine (Mg,Fe)2SiO4 or a magnesium iron silicate), the chemical formula becomes involved, as certain elements may substitute for another of similar size and charge.
-
The atoms within minerals are lined up in an orderly fashion, so that the characteristic crystalline structure is just an outward manifestation of the internal atomic arrangement.
What Are the Physical Properties of Minerals?
Identifying a mineral is a bit like playing detective. Minerals are identified by their physical properties. Physical properties are assessed by using observational skills. Observe Figure 3.2: how would you describe the mineral in this image?

Figure 3.2: Sulfur from Baja California, Mexico
You might describe this mineral as being yellow, with a shine and a distinct shape. Each description is actually a physical property (yellow = color, shiny = luster, shape = crystal form). As we learn more about minerals, it is important to note all available physical properties. Certain physical properties are referred to as diagnostic; this means they are useful in identifying a particular mineral by name. Those properties that are not diagnostic are still helpful, but often other properties are required for a proper identification. Color is a property that is frequently variable and should not be used as a diagnostic property. For example, quartz, an important rock-forming mineral, is available in a variety of colors; other properties would be necessary for an accurate identification. However, occasionally color can be helpful, as in the case of the mineral azurite. Azurite (from azure) is always blue in color.
Assessing the “importance” of one property over another, requires hours of practice with the minerals and their properties. This helps develop a mental mineral database, which can be used for future mineral identification. However, this database is only as good as the time and energy spent to develop it; geology majors, take note! Below are details regarding each of the physical properties to help identify minerals.
Mineral Properties: Visual Properties
Properties can be organized into three major categories: visual, testable, and special or unique properties.
This represents the outward color of the mineral and reflects atomic bonding or trace impurities. The outward color may be light, dark or anywhere in between. The color could be solid or mottled, and may even show iridescence (Figure 3.3).

Figure 3:3: The mineral labradorite, which displays iridescence.
This represents the minerals’ ability to transmit light. If light is easily passed through, and the mineral is see-through, it is described as being transparent. If light cannot pass through a mineral it is described as being opaque. If a mineral can pass light through, but is unable to be see-through, it is described as translucent. Certain minerals may have unique optical properties, like double refraction or fiber optics.
Luster refers to the appearance of the reflection of light from a mineral’s surface. Typically, there are two major luster categories, metallic, and non-metallic. Minerals with a metallic luster are typically opaque and appear as tarnished metal to a polished metal surface (Figure 3.4, left). A non-metallic luster encapsulates many possible appearances, from earthy to pearly, waxy to dull, glassy to silky (Figure 3.4, right). Submetallic lusters are also possible, and more subtle than a true metallic luster.

Figure 3:4: Left: A gold nugget from California displaying a metallic luster. Right: Botryoidal nephrite jade from California displaying a non-metallic, waxy luster
By definition all minerals are crystals, however some minerals will have a distinct crystal shape (Figure 3.5). This shape is determined by the minerals’ growth space. If a mineral has time and space to develop it forms crystals faces. These faces intersect to form a distinct crystal shape. Perfect crystals are rare, and are often what we associate with minerals viewed in a museum. Should a mineral not have space to grow, then no distinct crystal shape will develop. A mineral with no distinct geometry is given the term massive or compact.

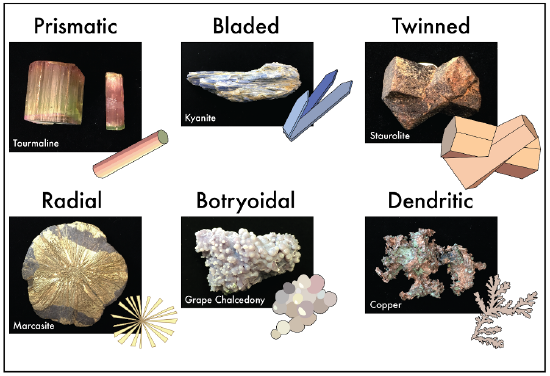
Figure 3:5: Various crystal habits and representative mineral examples
Mineral Properties: Testable Properties
Heft is related to specific gravity, or density, of the mineral. When geologists are in the field they are unable to take with them the equipment necessary to calculate specific gravity. Instead the heft test is preferred. The mineral is held in the palm of the hand, and the overall heaviness of the mineral is assessed. Does the mineral feel very heavy for its size? Very light? The chemistry of certain minerals, for example the metal bearing minerals, will often result in a high heft and feel heavy in your palm. Other minerals, like kaolinite or sulfur will feel relatively lighter in heft for their size.
Specific gravity is the ratio of a mineral’s weight to the weight of an equal volume of water. A mineral with a specific gravity of 2 would weigh twice as much as water. Most minerals are heavier than water, and the average specific gravity for all minerals is approximately 2.7. Some minerals are quite heavy, such as pyrite with a specific gravity of 4.9-5.2, native copper, with a specific gravity of 8.8-9.0, and native gold at 19.3, which makes panning useful for gold, as the heavy mineral stays behind as you wash material out of the pan.
This is how the mineral feels beneath your fingertips. We want to move away from the term “texture”, as this term will have a distinct meaning as we move into our rock units. Minerals may have a distinct feel: for example, olivine typically has a gritty or granular feel, whereas talc often feels greasy or soapy.

Figure 3.6: Streak plate with Pyrite (left) and Rhodochrosite (right) and their characteristic streak color.
Streak is the color of the mineral left behind on an unglazed porcelain plate, or a streak plate (Figure 3.6). A mineral’s streak color may be different from its outward color. Many minerals have a white streak, which is not particularly diagnostic. However, a mineral with a colored streak – black, blue, gray, green, or red – is often diagnostic. If you are ever unsure of the streak color, wipe your finger along the streak plate; a mineral with a white streak will leave a white powder behind that will rub on your finger. If your instructor provides both a white and black streak plate, try streaking the mineral on both plates. Recall, if a mineral is harder than the ceramic streak plate, you will see a porcelain powder from the plate and not from the mineral.
Tenacity refers to the way a mineral resists breakage and represents the brittleness, flexibility or malleability of a mineral. It is different from a mineral's hardness. If a mineral shatters like glass, it is said to be brittle, like quartz, while minerals that can be hammered are malleable, like copper or gold. Minerals may be elastic, in which they are flexible and bend like a plastic comb but return to their original shape, like the micas. Sectile minerals are soft like wax, and can be separated with a knife, like gypsum.
Hardness refers to the resistance of a mineral to being scratched by a different mineral or other material and is a product of the strength of the bonds between the atoms of a mineral. Whatever substance does the scratching is harder; the item scratched is softer. Hardness is based on a scale of 1 to 10, the Mohs Hardness Scale, created by a mineralogist named Friedrich Mohs (Table 3.1).
| Hardness number | Mineral | Test kit item |
|---|---|---|
| 10 (hardest) | Diamond | |
| 9 | Corundum | |
| 8 | Topaz | 8.5 - Masonry drill bit |
| 7 | Quartz | |
| 6 | Feldspar | 6.5 - Steel nail |
| 5 | Apatite | 5.5 - Glass plate or knife blade |
| 4 | Fluorite | |
| 3 | Calcite | 3.5 - Copper |
| 2 | Gypsum | 2.5 - Fingernail |
| 1 (softest) | Talc |
The Mohs’ scale lists ten minerals in order of relative hardness. Each mineral on the scale can scratch a mineral of a lower number. Your mineral kits will likely contain several items of a known hardness. The glass plate and steel nail have a hardness of 5.5, a copper penny, plate or sheet has a hardness of 3, and your fingernail has a hardness of 2.5. A mineral scratched by your fingernail is relatively soft, and its assessed hardness would be <2.5. When trying to scratch a surface, use force, but be cautious with the glass plate. ALWAYS lay the glass plate on a flat surface rather than holding it in your hand.
Occasionally mineral powder is left behind when a soft mineral scratches a harder surface, so feel for the groove or scratch. Materials of similar hardness may have difficulty scratching each other. For example, hornblende (amphibole) typically has a hardness right around glass, so it may or may not leave a scratch on the glass surface.
Minerals, even those with high hardness, can break. Typically, minerals break in one of two ways. A mineral can break with fracture or with cleavage. The type of breakage is dependent on the chemical bonds that are present at the atomic level of the mineral. There are many types of chemical bonds and forces that bind molecules together. The two most basic types of bonds are characterized as either ionic or covalent. In ionic bonding, electrons are being transferred from one atom to another in an ionic bond (Figure 3.7, left). These bonds are important to the formation of many minerals and are very strong. In contrast, covalent bonds share electrons and are also often strong bonds (Figure 3.7, right). Most minerals are characterized by ionic bonds, covalent bonds, or a combination of the two, but there are other types of bonds that are important in minerals, including metallic bonds and weaker electrostatic forces (hydrogen or Van der Waals bonds). Metallic elements have outer electrons that are relatively loosely held. When bonds between such atoms are formed, these electrons can move freely from one atom to another. A metal can thus be thought of as an array of positively charged atomic nuclei immersed in a sea of mobile electrons. This feature accounts for two very important properties of metals: their electrical conductivity and their malleability. Molecules that are bonded ionically or covalently can also have other weaker electrostatic forces holding them together. Examples of this are the force holding graphite sheets together and the attraction between water molecules.
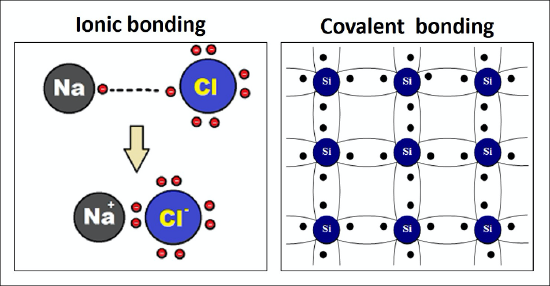
Figure 3:7: An example of ionic bonding in sodium chloride (NaCl) (left); an example of silicon covalent bonding (right)
Recall, all minerals are characterized by a specific three-dimensional pattern known as a lattice or crystal structure. These structures range from the simple cubic pattern of halite (NaCl), to the very complex patterns of some silicate minerals. Two minerals may have the same composition, but very different crystal structures and properties are known as polymorphs. Graphite and diamond, for example, are both composed only of carbon, but while diamond is the hardest substance known, graphite is softer than paper. Mineral lattices have important implications for mineral properties. Lattices also determine the shape that mineral crystals grow in and how they break. For example, the right angles in the lattice of the mineral halite influence both the shape of its crystals (typically cubic), and the way those crystals break.
A mineral which contains chemical bonds that are equal, and the same throughout, will not have a pattern of breakage and is said to exhibit fracture. This results in breakage that looks like when you break a glass or plate at home. Fracture surfaces are commonly uneven or conchoidal, a ribbed, smoothly curved surface similar to broken glass.
A mineral which contains chemical bonds that are unequal, and are different throughout, will result in a pattern of breakage called cleavage. This results in breakage with a shape that looks like itself, every time the mineral is broken. As minerals are broken (such as with a rock hammer), any cleavage planes will result in flat surfaces parallel to the directions of weakness within the crystal, these are the zones of weakness.
A mineral may have one or more cleavage planes (Figure 3.8). Planes that are parallel are considered the same direction of cleavage and should only count as one.
-
One direction of cleavage is termed basal or sheet cleavage. Minerals that display this cleavage will break off in flat sheets.
-
Two directions of cleavage is termed prismatic.
-
Three directions of cleavage at 90° is termed cubic.
-
Three directions of cleavage NOT at 90° is termed rhombic.
-
Four directions of cleavage is termed octahedral.
When 2 or more cleavage planes are present, it is important to pay attention to the angle of the cleavage planes. To determine the angle of cleavage, look at the intersection of cleavage planes. Commonly, cleavage planes will intersect at 90° (right angles) or 60°/120°. It can be challenging to distinguish cleavage from crystal form, particularly in imperfect samples. However, crystal form occurs as a mineral grows, while cleavage develops as a mineral breaks.
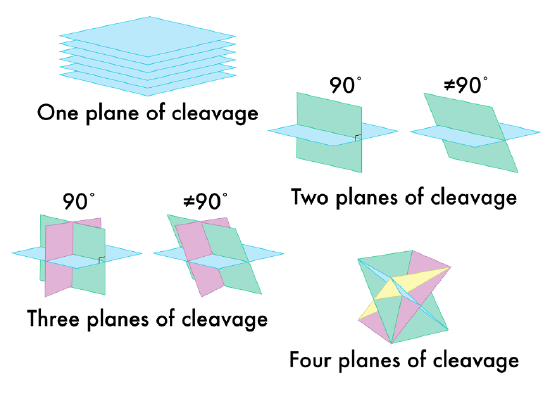
Figure 3:8: Various cleavage planes and directions
Special or Unique Properties
Effervescence
Some minerals react when dilute hydrochloric acid (HCl) is placed on them (Figure 3.9). When hydrochloric acid is introduced to a mineral, like calcite, a carbonate mineral, a chemical reaction occurs. The mineral effervesces, fizzes or bubbles and releases carbon dioxide gas. When you test a mineral with acid, be cautious and use just a drop of the acid. Use your magnifying glass to look closely for bubbles. The acid is very dilute and will not burn your skin or clothing, but you should always wash your hands after use. Also make sure that you rinse with water and wipe off the acid from the minerals that you test. Occasionally, vinegar can also be used, but larger quantities are necessary and the reaction becomes muted.

Magnetism
Certain metal bearing minerals will be attracted to a magnet. Magnetite, an iron oxide, is an important mineral in sea floor striping; stripes oriented parallel to mid-ocean ridges, which will be discussed more in Plate Tectonics.
Taste
Some minerals have distinct tastes, which can be useful in determining the mineral name. Commonly, the halides can be tasted and, in the case of halite, taste familiar (it is common table salt!). Other salts will have more bitter or sweet flavors depending on the types. However, YOU SHOULD NEVER TASTE UNKNOWN MINERALS. Tasting unknown minerals is an unsafe practice, as there are many minerals that you should not ingest. Only taste minerals cleared by your instructor.
Smell
Commonly, sulfur bearing minerals have a distinct rotten egg odor. This can be a clue to the chemical composition of the mineral, which points to specific mineral groups and eliminates others. When smelling minerals do not smell the mineral directly, instead waft the smell to your nose.
Fluorescence
Fluorescent minerals are those that emit visible light when activated by invisible ultraviolet light (UV), X-rays and/or electron beams. Certain electrons in the mineral absorb the energy from these sources and jump to a higher energy state, this results in many of the dayglo colors (Figure 3.10).

Surface Markings
At first glance all the minerals within the feldspar group look similar. However, potassium feldspar (K-spar), and plagioclase feldspar (varieties include labradorite and albite) do have distinct differences between them. While all feldspars have two cleavage planes that intersect at 90° and typically demonstrate vitreous luster and occur in opaque shades of white to gray to pink to very dark gray. A major difference between K-spar and plagioclase are the appearance of markings on the cleavage faces. Plagioclase feldspars will typically show tiny grooves, or striations on one cleavage face (Figure 3.11, left). Potassium feldspar will exhibit exsolution lamellae, wavy lines look like wood grain, instead of striations (Figure 3.11, right).

Mineral Groups
Most minerals are made up of a cation (a positively charged ion) or several cations and an anion (a negatively charged ion). For example, in the mineral hematite (Fe2O3), the cation is Fe3+ (iron) and the anion is O2– (oxygen). We group minerals into classes on the basis of their predominant anion or anion group. Groups include the oxides, sulfides, carbonates, silicates, and others. The silicates are by far the predominant group in terms of their abundance within the crust and mantle. Examples of common minerals from the different groups are given in Table 3.2.
| Mineral Group | Mineral Examples |
|---|---|
| Oxides | Hematite, corundum, water ice |
| Sulfides | Galena, pyrite, chalcopyrite |
| Sulfates | Gypsum, barite |
| Halides | Fluorite, halite |
| Carbonates | Calcite, dolomite |
| Phosphates | Apatite, Turquoise |
| Silicates | Quartz, feldspar, olivine |
| Native minerals | Gold, diamond, graphite, sulfur, copper |
What Do These Mineral Groups Mean?
Oxide minerals have oxygen (O2–) as their anion, but they exclude those with oxygen complexes such as carbonate (CO32–), sulfate (SO42–), and silicate (SiO44–). The most important oxides are the iron oxides hematite and magnetite (Fe2O3 and Fe3O4, respectively). Both of these are important ores of iron. Corundum (Al2O3) is an abrasive, but can also be a gemstone in its ruby and sapphire varieties. If the oxygen is also combined with hydrogen to form the hydroxyl anion (OH–) the mineral is known as an hydroxide. Some important hydroxides are limonite and bauxite, which are ores of iron and aluminum respectively. Frozen water (H2O) is a mineral (an oxide), but liquid water is not because it doesn’t have a regular lattice.
Sulfides are minerals with the S2- anion, and they include galena (PbS), sphalerite (ZnS), chalcopyrite (CuFeS2), and molybdenite (MoS2), which are the most important ores of lead, zinc, copper, and molybdenum, respectively. Some other sulfide minerals are pyrite (FeS2), bornite (Cu5FeS4), stibnite (Sb2S3), and arsenopyrite (FeAsS).
Sulfates are minerals with the SO42- anion, and these include anhydrite (CaSO4) and its cousin gypsum (CaSO4.2H2O) and the sulfates of barium and strontium: barite (BaSO4) and celestite (SrSO4). In all of these minerals, the cation has a +2 charge, which balances the –2 charge on the sulfate ion.
The halides are so named because the anions include the halogen elements chlorine, fluorine, bromine, etc. Examples are halite (NaCl), cryolite (Na3AlF6), and fluorite (CaF2).
The carbonates include minerals in which the anion is the CO32- complex. The carbonate combines with +2 cations to form minerals such as calcite (CaCO3), magnesite (MgCO3), dolomite ((Ca,Mg)CO3), and siderite (FeCO3). The copper minerals malachite and azurite are also carbonates.
In phosphate minerals, the anion is the PO43- complex. An important phosphate mineral is apatite (Ca5(PO4)3(OH)), which is what your teeth are made of.
The all-important silicate minerals include the elements silicon and oxygen in varying proportions ranging from Si:O2 to Si:O4.
Native element minerals are single-element minerals, such as gold, copper, sulfur, and graphite.
Attributions
- Figure 3.1: “Building Materials and Minerals” (Public Domain; USGS)
- Figure 3.2: “Sulfur from Baja California, Mexico” (CC-BY 2.0; James St. John via Flickr)
-
Figure 3.3: “Volga Blue Granite” (CC-BY 2.0; James St. John via Flickr)
-
Figure 3.4: Derivative of “Gold Nugget” (CC-BY 2.0; James St. John via Flickr) and “Botryoidal Nephrite Jade” (CC-BY 2.0; James St. John via Flickr) by Chloe Branciforte
-
Figure 3.5: “Crystal Habits” (CC-BY 4.0; Emily Haddad, own work)
-
Figure 3.6: “Streak Plate with Pyrite and Rhodochrosite” (CC-BY-SA 3.0; Ra’ike via Wikipedia)
-
Table 3.1: “Mohs Hardness Scale” (CC-BY 4.0; Chloe Branciforte, own work)
-
Figure 3.7: Derivative of “Ionic Bonding” (CC-BY-SA 3.0; Rhannosh via Wikimedia Commons) and “Silicon Covalent Bond” (Public Domain; Hermitage17 via Wikimedia Commons) by Chloe Branciforte
-
Figure 3.8: “Mineral Cleavage Planes” (CC-BY 4.0; Emily Haddad, own work)
-
Figure 3.9: “Effervescence” (CC-BY 4.0; Chloe Branciforte, own work)
-
Figure 3.10: “Fluorescent Minerals” (CC-BY 4.0; Chloe Branciforte, own work)
-
Figure 3.11: “Feldspar Markings” (CC-BY 4.0; Chloe Branciforte, own work)
-
Table 3.2: “Mineral Groups” (CC-BY 4.0; Chloe Branciforte, own work)


