11.3: Soil Properties
- Page ID
- 16124
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Soils can be enormously complex systems of organic and inorganic components. Here, we'll concentrate on a few of the most significant properties, texture, structure, color, and chemistry

Soil Texture
Soil texture refers to the relative proportion of sand, silt and clay size particles in a sample of soil. Clay size particles are the smallest being less than .002 mm in size. Silt is a medium size particle falling between .002 and .05 mm in size. The largest particle is sand with diameters between .05 for fine sand to 2.0 mm for very coarse sand. Soils that are dominated by clay are called fine textured soils while those dominated by larger particles are referred to as coarse textured soils. Soil scientists group soil textures into soil texture classes. A soil texture triangle is used to classify the texture class (see below).
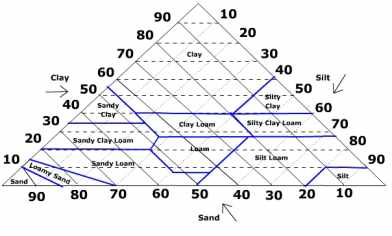
The sides of the soil texture triangle are scaled for the percentages of sand, silt, and clay. Clay percentages on the left side of the triangle are read from left to right across the triangle (dashed lines). Silt runs from the top to the bottom along the right side and is read from the upper right to lower left (light, dotted lines). The percentage of sand increases from right to left along the base of the triangle. Sand is read from the lower right towards the upper left portion of the triangle (bold, solid lines). The boundaries of the soil texture classes are highlighted in blue. The intersection of the three sizes on the triangle give the texture class. For instance, if you have a soil with 20% clay, 60% silt, and 20% sand it falls in the "silt loam" class.
What is the texture class for a soil having 45% clay, 45% silt, and 10% sand?
Slit Loam, Clay, Silty Clay, or Silty Clay Loam?
- Answer
-
Silty Clay
Soil texture effects many other properties like structure, chemistry, and most notably, soil porosity, and permeability. Soil porosity refers to the amount of pore, or open space between soil particles. Pores are created by the contacts made between irregular shaped soil particles. Fine textured soil has more pore space than coarse textured because you can pack more small particles into a unit volume than larger ones. More particles in a unit volume creates more contacts between the irregular shaped surfaces and hence more pore space. As a result, fine textured clay soils hold more water than coarse textured sandy soils. Permeability is the degree of connectivity between soil pores. A highly permeable soil is one in which water runs through it quite readily. Coarse textured soils tend to have large, well-connected pore spaces and hence high permeability.
Soil Structure
Soil structure is the way soil particles aggregate together into what are called peds. Peds come in a variety of shapes depending on the texture, composition, and environment.
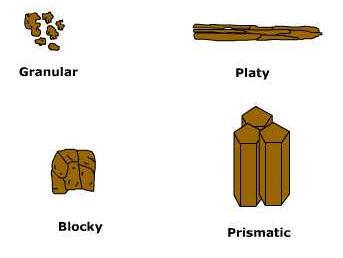
Granular, or crumb structures, look like cookie crumbs. They tend to form an open structure that allows water and air to penetrate the soil. Platy structure looks like stacks of dinner plates overlaying one another. Platy structure tends to impede the downward movement of water and plant roots through the soil. Therefore, open structures tend to be better agricultural soils.
Bulk density of a soil is the mass per unit volume including the pore space. Bulk density increases with clay content and is considered a measure of the compactness of the soil. The greater the bulk density, the more compact the soil. Compact soils have low permeability, inhibiting the movement of water. The use of heavy agricultural equipment can cause compaction of soil, especially in wet clay soil. Soil compaction results in reduced infiltration and increase runoff and erosion.
Soil Chemistry
As plant material dies and decays it adds organic matter in the form of humus to the soil. Humus improves soil moisture retention while affecting soil chemistry. Cations such as calcium, magnesium, sodium, and potassium are attracted and held to humus. These cations are rather weakly held to the humus and can be replaced by metallic ions like iron and aluminum, releasing them into the soil for plants to use. Soils with the ability to absorb and retain exchangeable cations have a high cation-exchange capacity. Soils with a high cation-exchange capacity are more fertile than those with a low exchange capacity.
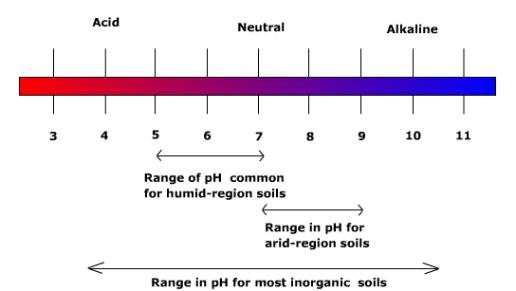
Hydrogen ion concentration in the soil is measured in terms of the pH scale. Soil pH ranges from 3 to 10. Pure water has a pH of 7 which is considered neutral, pH values greater than seven are considered basic or alkaline, below seven acidic. Most good agricultural soils have a pH between 5 and 7. Though acidic soils pose a problem for agriculture due to their lack of nutrients, alkaline soils can pose a problem as well. Alkaline soils may contain appreciable amounts of sodium that exceed the tolerances of plants, contribute to high bulk density and poor soil structure. Alkaline soils are common in semiarid regions.