11.2: Agents of Metamorphism
- Page ID
- 5664
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Pressure
All rocks beneath the surface of the earth experience an increase in pressure due to the weight of the overlying sediment and rock layers and with increasing depth, there is a corresponding increase in pressure. This increased pressure does not necessarily cause a rock to become metamorphic, because this particular pressure is typically equal in all directions and is known as lithostatic pressure. Lithostatic pressure is similar to hydrostatic pressure, such as the pressure on the eardrums a swimmer will experience as he or she dives deep in the water. Lithostatic pressure on rocks below the earth’s surface may have a change in overall rock volume, but will not cause a change in the shape. An example of decreasing volume due to lithostatic pressure would be a closer packing of clasts and reduction of pore space within a clastic sedimentary rock. But what if the pressure on a rock is unequal, and the rocks become squeezed in one direction more than another direction? This is known as differential pressure, and it can result in a significant change in the appearance of a rock. Figure 11.1 demonstrates how a mineral can change shape due to differential pressure, in this case with the greatest pressures from the top and bottom (as demonstrated by the large gray arrows). Two initially rounded mineral grains (Figure 11.1A) within a sedimentary rock are experiencing the greatest amount of pressure at the contact between the grains (see red arrows in the figure), and the bonds linking the atoms in this grain will break. The atoms will migrate into the area of lesser pressure and reform a bond with other atoms in the mineral grain (Figure 11.1B). As a result, the grains have a flattened shape that is perpendicular to the direction of greatest pressure (Figure 11.1C).
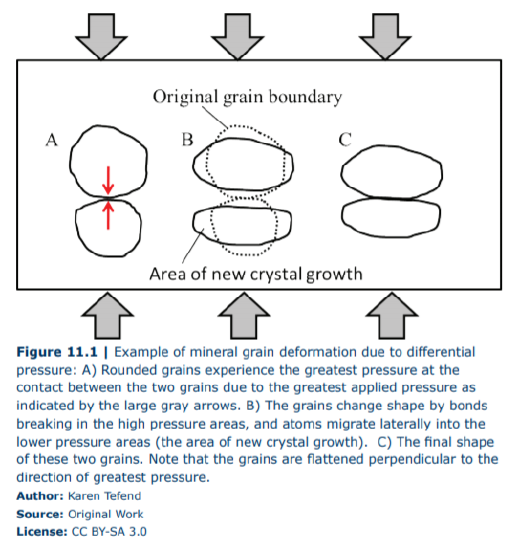
Figure 11.1 only shows the deformation of two grains; imagine that this is happening to all of the grains in the sedimentary rock, or to all of the phenocrysts (crystals) in an igneous rock. In that case, you will end up with the entire rock having minerals aligned in a certain direction, all by the breaking of bonds between atoms in a mineral, and reforming (recrystallizing) in the lower pressure areas among the grains or crystals in the rock. The end result is a rock with a metamorphic pattern called a foliation. Metamorphic foliations are the patterns seen in a rock that has experienced differential pressure; these foliations may be fairly flat or have a wavy appearance possibly due to more than one direction of greatest pressure. Some rocks may also develop what is called a lineation, which can be formed by an elongation of minerals that form a linear feature through the rock. To understand the difference between a foliation and a lineation, let us use some food analogies: a stack of pancakes demonstrates a foliation in your breakfast food, with each pancake layer representing flattened minerals. If you look at the top of the pancakes, you will not see a pattern, but if you view the stack of pancakes from the side, or cut through the stack with your knife in any orientation other than parallel to the pancake layers, you will see the layering or foliation. However, if within that pancake stack, there existed a slice of bacon (yum), the bacon would be the lineation in your breakfast “rock”, and you may or may not see it when you cut through the pancake stack. We will discuss foliations, and the different types of foliations, in a later section of this chapter.
Temperature
Probably the most common cause of metamorphism is a change in temperature. Often times metamorphism involves both an increase in temperature along with the pressure changes as described in the above section. Higher temperatures are often associated with metamorphism due to chemically reactive fluids (which we will discuss in the next section). The broad classification for metamorphism into low, medium and high grades of metamorphic change exists mainly due to temperature conditions; this will also be discussed in a later section.
Higher temperatures increase the vibrational energy between the bonds linking atoms in the mineral structure, making it easier for bonds to be broken in order for the recrystallization of the minerals into new crystal shapes and sometimes the development of foliations and lineations as described in the previous section. However, recrystallization can be due to just temperature changes without any differential pressure conditions, and when temperatures are increased, there can be a corresponding increase in mineral sizes as initially small minerals become fused into larger crystals. This fusing of numerous smaller mineral sizes into fewer and larger mineral sizes is known as annealing in metallurgy. However, for metamorphic rocks, it is still referred to as recrystallization.
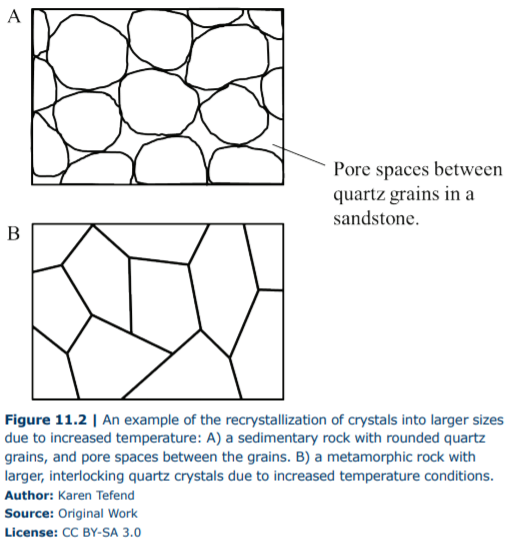
In order to understand why increasing temperatures lead to increased grain sizes, we need to again address stability. In general, a mineral grain or crystal is most stable when it has a low surface area to volume ratio, therefore large grains are more stable than small grains because increasing the grain size results in a greater increase in volume as opposed to a smaller increase in the surface area. Why does stability matter? Because that is why the rocks we are concerned with in this chapter are changing; rocks become unstable when their environment changes, and by a recrystallization process (metamorphism), they can return to a stable form once again. Figure 11.2 demonstrates the recrystallization process in sedimentary rocks in response to elevated temperature. In this example, the original grains are smaller and rounded, but recrystallization resulted in larger grains that are interlocking; the pore spaces are gone and instead larger crystals exist.
In addition to increased grain size with increased temperature, occasionally a new mineral forms during metamorphism. These new minerals form at certain temperatures and are called index minerals, which can be used to determine the temperature of metamorphism. Index minerals will be covered in more detail in a later section.
Chemically Reactive Fluids
The phrase chemically reactive refers to the dissolved ions in a fluid phase that may react with minerals in a rock; these ions may take the place of some of the atoms in the mineral’s structure, which may lead to a significant change in the chemical composition of a rock. Sometimes these fluids are quite hot, especially if they are fluids released from a nearby magma body that is crystallizing while cooling. Metamorphism due to such fluids is known as hydrothermal metamorphism.


