Oxygen Isotopes and Chemostratigraphy
- Page ID
- 10898
What is Chemostratigraphy?
Edited by Colin Boisvert, notes in blue
Chemostratigraphy Shouldn't this be in bold? is the study of the trace element and isotopic variability of sedimentary layers and is often used in conjunction with other types of stratigraphic analysis such as biostratigraphy, lithostratigraphy, or chronostratigraphy. It concerns itself with correlating geochemical changes with a geological event or age, a process known as fingerprinting Bolden. When we observe that a particular trace element or isotope responds to a certain process, we can use those observed changes to interpret a geologic event. When interpreted this way, we call that chemical a proxy for a given process. For example, oxygen isotopes respond to changes in temperature and are a common proxy for climate change in the past. By observing how oxygen isotopes vary in a stratigraphic sequence over time, one can infer what the climate in that region may have been like over that time period (1).
In geochemistry, many proxies are not measured by themselves as a total quantity but instead in relation to another relevant trace element or isotope as a ratio. For example, oxygen isotopes are measured as the abundance of one isotope to another: 18O/16O. This means that in order to interpret geologic events, we look at relative changes in one element or isotope when compared to another across a stratigraphic sequence (1, 2).
I would add an initial figure and table here to illustrate what you mean by oxygen demonstrates a proxy for paleoclimates. citing a paper and using a figure from it to show the change in temp over a period of time should do the trick like this paper Bohaty and Zachos 2003 Significant Southern Ocean warming event in the late middle Eocene. The 2 column table will demonstrate a list of 4-5 chemical proxies you can find and their relative use. ex. oxygen for paleoclimates,carbon for photosynthesis, etc... just for comparision can other proxies be used for paleoclimates?
Oxygen Isotopes
Why would we want to study oxygen isotopes specifically as a chemostratigraphic tool? Fundamentally, oxygen stable isotopes as a proxy respond to temperature variations brought about by climate change. Meaning that if we study oxygen isotopes, we can gain some insight into climatic controls over time. They are usually studied in some hydrologic context and can be observed in most naturally-occurring substances that contain oxygen. These substances include, but are not limited to: carbonate shells from marine and terrestrial sediments, calcite deposits, ice sheets(specifically Ice cores from these sheets), and liquid water.
We trace oxygen isotopes by looking at the ratio of 18O to 16O. Over 99% of the oxygen on Earth is 16O, with most of the remainder being 18O (1,2). Since there is significantly less 18O on Earth, we can compare subtle fluctuations in 18O relative to 16O to get a sense of what climatic processes were happening at some point or region over a certain amount of time. Give a short sentence on why there is less 18O compared to 16O and which does evaporation prefer when sucking water out of the ocean.
It is worth noting that oxygen isotopes are not the only proxy with which we can study climate change. Other proxies, such as strontium isotopes, carbon isotopes, or trace elements, also describe different processes and have different drivers. Since oxygen isotopes tend to respond to temperature variability, they can give us a picture of basic environmental and/or climatic changes over time. Coupling oxygen isotope records with other proxies gives that picture more detail.
As stated above add the table to illustrate this last paragraph.
Measuring Oxygen Isotopes
18O/16O values are reported relative to international standards usually VSMOW. By using a standard, this provides a reference point for the observed values and gives context to the observer. Values for aqueous oxygen isotopes, sourced from solid, liquid, and gaseous phases, are reported against Vienna Standard Mean Ocean Water (VSMOW). Values for carbonaceous oxygen isotopes, such as those from seashells or calcite deposits, are reported against Vienna Pee Dee Belemite (VPDB). When reporting relative to a standard, these proxies use the following delta notation (1):
*(Equation from ref. 2)
Where,
(18O/16O)sample = the value of your sample
(18O/16O)standard = the reference value of your standard (either VSMOW or VPDB)
d18O = the notation used to represent the value of your sample relative to the standard (called "delta 18 O")
It is important to note that d18O is reported in ‰, or “per mil”, hence the multiple of 1000 included in the calculation. ‰ simply means “parts per thousand”, much like how % means “parts per hundred” or “per cent”.
The Water Cycle and Oxygen Isotopes
To understand how we can trace oxygen isotopes and interpret their variability in these proxies, we must review the water cycle. In general, water evaporates out of the oceans, condenses into clouds, precipitates over land as rain and snow, forms river runoff, and is deposited back into the ocean where the cycle restarts (Fig. 1). At each of these stages, oxygen isotopes in water molecules respond, or sort themselves, in a way that we can measure. As mentioned before, oxygen isotopes respond to temperature, and they do so in a way sorting themselves, with respect to their relative masses.

Figure 1. A simplified water cycle, emphasizing evaporation, precipitation, and runoff (3).
We will start with water evaporating from the ocean. Since 16O is more lightweight than 18O, it will have a greater tendency to evaporate out of the ocean than 18O will. This means that 18O/16O in the ocean goes up (because more 18O stays in the ocean than leaves it) and 18O/16O of the water vapor goes down (because 18O generally stays in the ocean).
Once a cloud forms and moves over land, 18O has a tendency to become more mobile. Since it is heavier, it will tend to condense, form raindrops, and fall to the ground relatively more frequently than 16O will. This means that rain will tend to have a higher 18O/16O than the cloud that it came from. In this sense, 18O falls out of clouds more frequently than 16O does because it is literally heavier. This is more pronounced with proximity to the ocean, meaning that places near coastlines will have higher d18O when compared to places further inland.
Once a cloud reaches a cold enough area to produce snow, that snow has a much lower d18O value when compared to the original, evaporated ocean water because so much of the source 18O has already fallen out. This usually means that snow, and therefore ice sheets, contain a disproportionately large amount of 16O (the lighter isotope) when compared to 18O (the heavier isotope) (2).
River runoff usually has an “in-between” value, since it can be a combination of snow melt from upstream, rainwater, and input water from other sources.
Consequently, this all means that in order to move more 18O out of the ocean and onto any location on land, you must put more energy into the ocean by increasing the temperature. This means that, for example, proxies in marine sediments showing decreased d18O is a sign of a warm, possibly ice-free period (2). This implies that at the time those sediments formed, there was more 18O throughout the hydrologic cycle in general and that much of the 16O-rich ice had melted and re-integrated with the oceans. If those same proxies showed higher d18O, then that would be a sign of a cold period, possibly an ice age, and would have the opposite implications (2). This is all also true for many terrestrial proxies, but with reversed d18O values corresponding to warm and cold events (1). For reference, Figure 2 shows the typical d18O values for different ice and water reservoirs (2). Careful with these last 3 paragraphs the way the 18O/16O ratio operates between the evaporation condensation cycle and glaciers is different while the ideas are somewhat connected and should be stated as such. These paragraphs give me the idea they are more linked. Since 16O is light it gets sucked into glaciers more leaving a heavier ocean during cold times and then when melt occurs the ratio goes back down but this is slightly different from the evaporation cycle ratios as learned in Gel 108. this may help
https://www.seas.harvard.edu/climate/eli/research/equable/isotope.html
Figure 2. Typical d18O values at different regions on Earth. Polar ice caps tend to have very negative d18O values, reflecting a higher concentration of 16O relative to 18O (2). In contrast, the deep ocean waters have slightly positive values, reflecting opposite concentrations. Both the negative values seen in ice and the positive values seen in the ocean are consistent with the idea that 18O tends to stay in the ocean unless enough energy is applied to move it out of the ocean.
Oxygen Isotopes as a Chemostratigraphic Tool
Given the natural range of d18O values shown in Figure 2, it is important to note that different types of proxies will produce different d18O values. For example, very positive values in marine sediments potentially describe the same geologic event as very negative values in polar ice cores. This section uses benthic foraminifera fossils (Fig. 3) from marine sediments as an example for how we construct d18O chemostratigraphic records for paleoclimates.
Figure 3. Examples of benthic foraminifera fossils (4).
Foraminifera (or “forams”, for short) are microscopic, single-celled organisms. There are planktonic varieties, which live within the uppermost water column, and benthic varieties, which live along the seafloor. Both kinds of forams take dissolved calcium and bicarbonate ions directly from the water column to build their calcium carbonate (CaCO3) shells. In doing so, they preserve the d18O signature of the surrounding water within their shells. When they die, their shells are buried and preserved in the sediment. By examining the d18O variability from layer to layer and cross-referencing with chronostratigraphy and/or age-dating, forams can be used to create a natural record of ocean d18O variability through time. Sure forams can record the delta18O record but why can they create a natural record of ocean variabity through time. (its because they are considered index fossils and species change rapidly over well studied short temporal ranges combined with the oxygen isotope record they provide to create a bigger picture of the paleoclimate of the ocean through time.
As previously discussed, d18O fundamentally responds to temperature variability. Likewise, the d18O preserved in foram shells also depends on surrounding temperatures and will show enrichment in 18O (more positive values) when the surrounding water is colder and depletion in 18O (less positive values) when the surrounding water is warmer. In other words, the shells reflect the composition of the water at the time the shells were formed and that composition gives us insight to how both the ocean environment and global climate dynamics may have changed over time (1).
Benthic forams are most often studied in d18O stratigraphy because they are easier to interpret d18O variability through time (Fig. 4). Since they live on the bottom sediment and have more limited mobility, their d18O signal theoretically responds only to temporal environmental changes. Planktonic forams are more likely to move around, causing their d18O signals to reflect more spatial variability. These forams may have been transported across some horizontal distance, giving a d18O signal that represents an unknown location. Additionally, since planktonic forams live at various depths within the water column, and temperature of the water can vary with depth, their d18O signal may actually reflect vertical spatial variability rather than environmental change over time. Studying benthic forams allows for location to be controlled so changes in d18O can reflect changes over time (2). Again, great paragraph but don't forget to mention why over time unless you have a consecutive series of species living that have constricted time windows to study the variation to build the bigger picture.
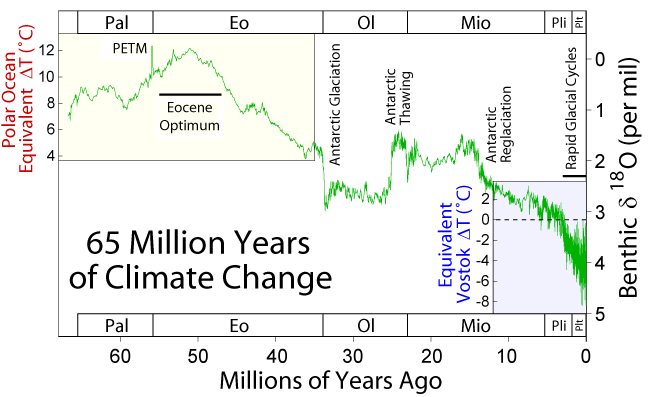
Figure 4. Climatic record of the Cenozoic as documented by d18O from benthic foraminifera. More negative values (toward the top of the chart) represent warmer periods. More positive values (toward the bottom of the chart) represent colder periods. Several dramatic shifts in d18O have been noted. The PETM (Paleocene-Eocene Thermal Maximum) represents a temporary(use rapid instead of temporary as this event lasted 170ka and temporary isn't a clear geologic term), intense warming period. The Antarctic Glaciation is marked by a permanent, positive shift at the end of the Eocene that cooled the Earth and caused the Antarctic ice sheet to form. At the end of the Oligocene, there was a negative shift that caused melting of the ice sheet. After about 10 million years of stability, a continuous positive shift occurred. The Antarctic ice sheet reformed and the Earth went through several glacial cycles (Ice ages)up to the present (5). I wouldn't italicize the figure caption hard to read. good figure though something like this for the top of the page.
Great start. Can you provide more information on why one would want to use d18O as a chemostratigraphic tool (noted, working on that)? Also, they reflect water d18O in addition to temperature. And in fact, temperatures vary significantly laterally, so the temperature dependence of d18O isn't the part that makes it a stratigraphic tool. It's the changes in seawater d18O due to ice volume that change ocean chemistry globally, making d18O useful as a stratigraphic tool. The temperature dependence is very useful for reconstructing ancient environments, though, when the water d18O changes can be accounted for as well (Great points. Temperature dependence is the driving factor, so that's important to include. This is partially addressed in the newly-added water cycle section. The next step is talking about how forams record isotopes in the water column and that will address how d18O is useful as a stratigraphic tool.). Section 3.3.1.3 of this chapter will be helpful: https://www.sciencedirect.com/scienc...24199682000030
good work cited, very clear
The page is very well done and thorugh, once the few extra explanations/ misunderstandings are worked out this page is ready to rock and relevant figures in appropriate places.
References:
(1) Ramkumar, Mu. Chemostratigraphy: Concepts, Techniques, and Applications. Elsevier publishing, 2015.
(2) Ruddiman, William F. Earth’s Climate: Past and Future. W. H. Freeman and Company, 2008.
(3) [I'm not really sure how to cite this one] https://encounteredu.com/multimedia/images/why-is-the-water-cycle-important-to-us
(4) [I'm not really sure how to cite this one] https://commons.wikimedia.org/wiki/File:Benthic_foraminifera.jpg
(5) [I'm not really sure how to cite this one] https://commons.wikimedia.org/wiki/File:65_Myr_Climate_Change.png

