7.3: Nucleation of Ice Crystals
- Page ID
- 9570
Ice cannot survive at temperatures above 0°C at normal atmospheric pressures. Below that temperature, ice crystals can exist in equilibrium with air that is supersaturated with respect to ice. The saturation curve for ice was plotted in Fig. 4.2, and is close to, but slightly below, the curve for supercooled liquid water.
There is a thermodynamic barrier to ice formation, analogous to the barrier for droplet growth. This barrier can be overcome with either very cold temperatures (colder than –40°C), high supersaturation in the air, or by the presence of ice nuclei.
7.3.1. Processes
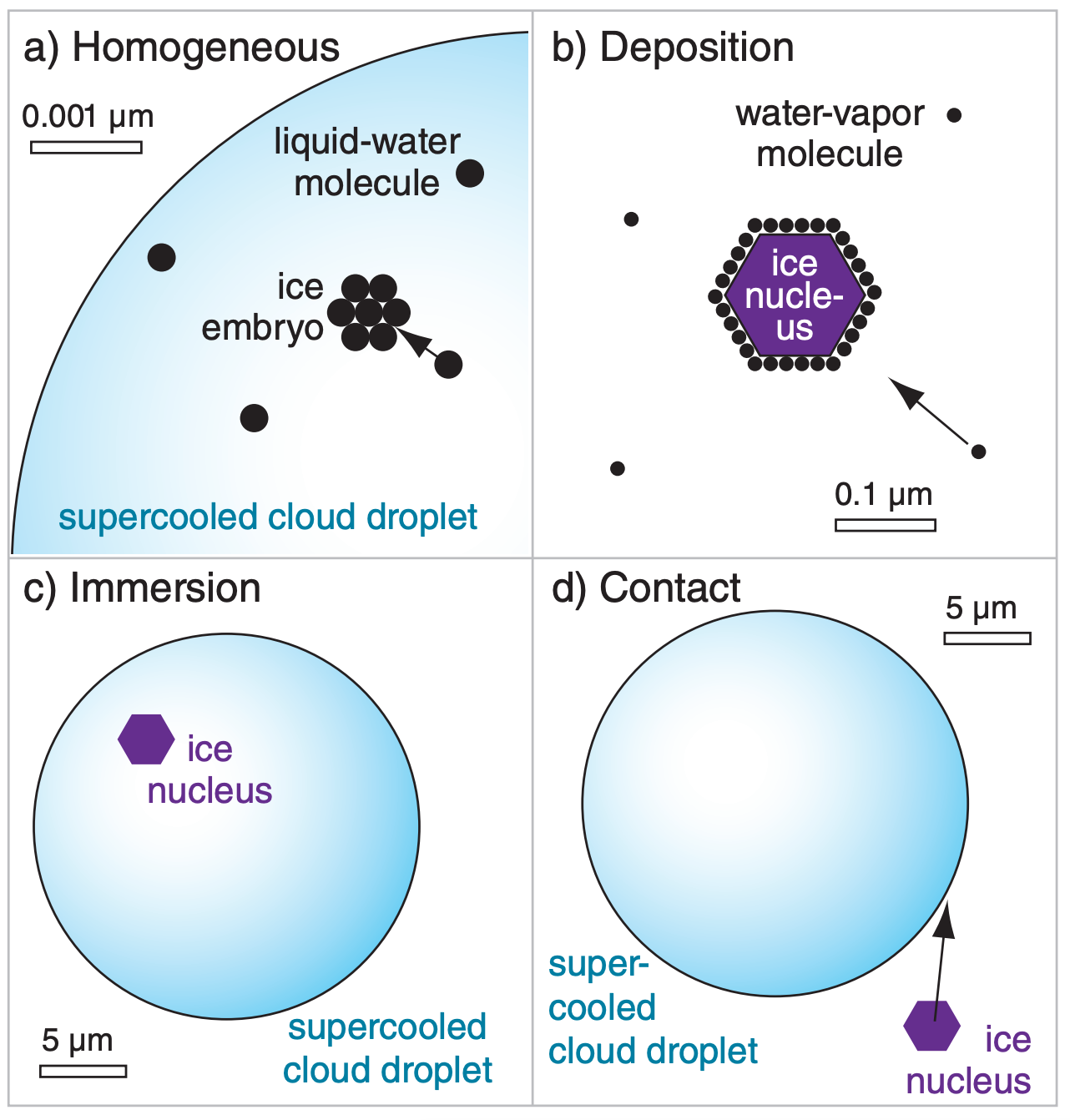
Homogeneous freezing nucleation is the name for the spontaneous freezing that occurs within supercooled liquid-water droplets as temperature decreases to near –40°C. No impurities are needed for this. Instead, ice embryos form by chance when clusters of water molecules happen to come together with the correct orientations (Fig. 7.9a). At –40°C, the cluster needs only about 250 molecules. At slightly warmer temperatures the critical embryo size is much larger, and thus less likely to occur. Because larger supercooled droplets contain greater numbers of molecules, they are more likely to form an ice embryo and freeze.
Heterogeneous freezing nucleation can occur a variety of ways (Fig. 7.9b-d), but all require an impurity (Table 7-2), which is generically called an ice nucleus. These processes are described next.
Deposition nucleation (Fig. 7.9b) occurs when water vapor deposits directly on a deposition nucleus. While the solute effect was important for liquid droplet nucleation, it does not apply to ice nucleation because salts are excluded from the ice-crystal lattice as water freezes. Thus, the size and crystal structure of the ice nucleus is more important. Deposition is unlikely on particles of size 0.1 µm or less. Colder temperatures and greater supersaturation increase deposition nucleation.
Immersion freezing (Fig. 7.9c) occurs for those liquid drops already containing an undissolved ice nucleus called a freezing nucleus. Each nucleus has a critical temperature below 0°C at which it triggers freezing. Thus, as external processes cause such a contaminated drop to cool, it can eventually reach the critical temperature and freeze.
Larger drops contain more freezing nuclei, and have a greater chance of containing a nucleus that triggers freezing at warmer temperatures (although still below 0°C). To freeze half of the drops of radius R, the temperature must drop to T, given statistically by
\(\ \begin{align} T \approx T_{1}+T_{2} \cdot \ln \left(R / R_{o}\right)\tag{7.18}\end{align}\)
where Ro = 5 µm, T1 = 235 K, and T2 = 3 K.
Condensation freezing is a cross between deposition nucleation and immersion freezing. In this scenario, which occurs below 0°C, nuclei are more attractive as condensation nuclei than as deposition nuclei. Thus, supercooled liquid water starts to condense around the nucleus. However, this liquid water immediately freezes due to the immersionnucleation properties of the nucleus.
Contact freezing (Fig. 7.9d) occurs when an uncontaminated supercooled liquid drop happens to hit an external contact nucleus. If the droplet is cooler than the critical temperature of the ice nucleus, then it will freeze almost instantly. This also happens when (supercooled) freezing rain hits and instantly freezes on trees and power lines. Ice crystals in the air are also good contact nuclei for supercooled water.
| Table 7-2. Listed are the warmest ice nucleation threshold temperatures (°C) for substances that act as ice nuclei. Processes are: 1 contact freezing, 2 condensation freezing, 3 deposition, and 4 immersion freezing. (x = not a process for this substance.) | ||||
| Substance | Process | |||
| 1 | 2 | 3 | 4 | |
| silver iodide | –3 | –4 | –8 | –13 |
| cupric sulfide | –6 | x | –13 | –16 |
| lead iodide | –6 | –7 | –15 | x |
| cadmium iodide | –12 | x | –21 | x |
| metaldehyde | –3 | –2 | –10 | x |
| 1,5-dihydroxynaphlene | –6 | –6 | –12 | x |
| phloroglucinol | x | –5 | –9 | x |
| kaolinite | –5 | –10 | –19 | –32 |
Sample Application
What cloud temperature is needed to immersionfreeze half of the droplets of 100 µm radius?
Find the Answer
Given: R = 100 µm. Find: T = ? K
Use eq. (7.18)
T = 235K + (3K)·ln(100µm/5µm) = 244K = –29°C
Check: Units OK. Physics OK.
Exposition: Smaller droplets can remain unfrozen at much colder temperatures than larger drops, which are thus available to participate in the WBF precipitation growth process described later.
7.3.2. Ice Nuclei
Only substances with similar molecular structure as ice can serve as ice nuclei. Such substances are said to be epitaxial with ice.
Natural ice nuclei include fine particles of clay such as kaolinite stirred up from the soil by the wind. Certain bacteria and amino acids such as lleucine and l-tryptophan from plants also can nucleate ice. Combustion products from forest fires contain many ice nuclei. Also, ice crystals from one cloud can fall or blow into a different cloud, triggering continued ice formation.
Other substances have been manufactured specifically to seed clouds, to intentionally change the amount or type (rain vs. hail) of precipitation. Silver iodide has been a popular chemical for cold-cloud seeding. Other cold-cloud-seeding chemicals are lead iodide, cupric sulfide, phloroglucinol, and metaldehyde. Table 7-2 lists some ice nuclei and their critical temperatures. Because contact nucleation occurs at the warmest temperatures for most substances, it is the most likely process causing ice nucleation.
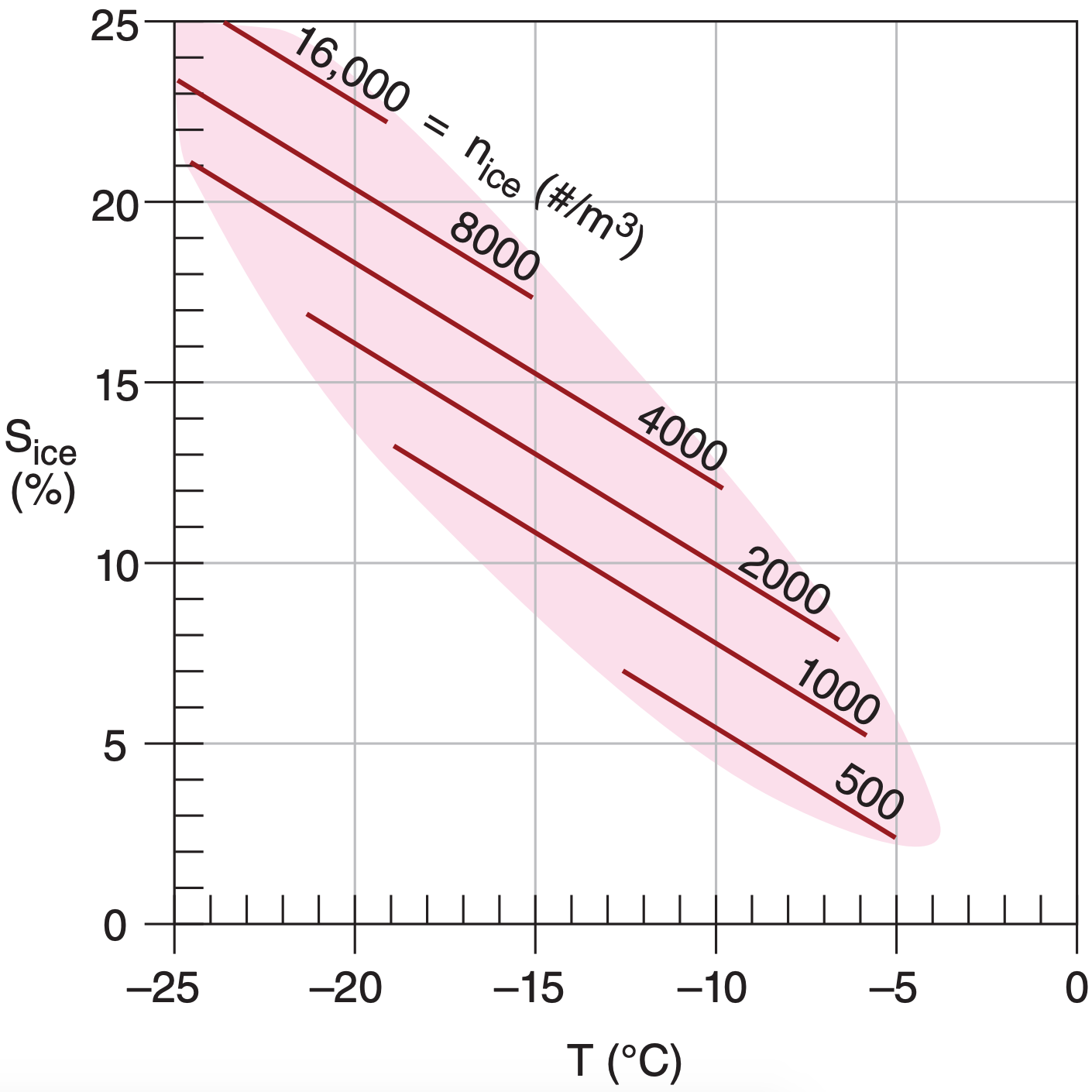
Air usually contains a mixture of ice nuclei having a variety of ice nucleation processes that become active at different temperatures and supersaturations. The number density nice of active ice nuclei per cubic meter of air is shown in Fig. 7.10.