7.8: The Thermal Structure of Glaciers
- Page ID
- 14340
The Thermal Structure of Glaciers
Processes of Energy Exchange at Glacier Boundaries
A glacier can gain or lose heat in several ways. It’s clear that these processes operate both at the surface of the glacier and at the base of the glacier. What are these processes, and what is their relative importance? Table 1 lists the ways heat can be gained or lost by a glacier at its surface.
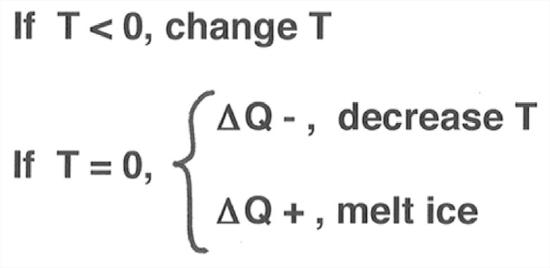
First of all, keep in mind that the combined effect of all of these processes depends on whether the glacier is at its melting point or below its melting point (Figure 7-25). Obviously, if the temperature of the glacier is below the melting point, then the effect of addition of heat is to raise the temperature and the effect of extraction of heat is to lower the temperature. But if the temperature of the glacier is at the melting point, then addition of heat serves to melt glacier ice. Of course, extraction of heat when the glacier is at the melting point lowers the temperature below the melting point.
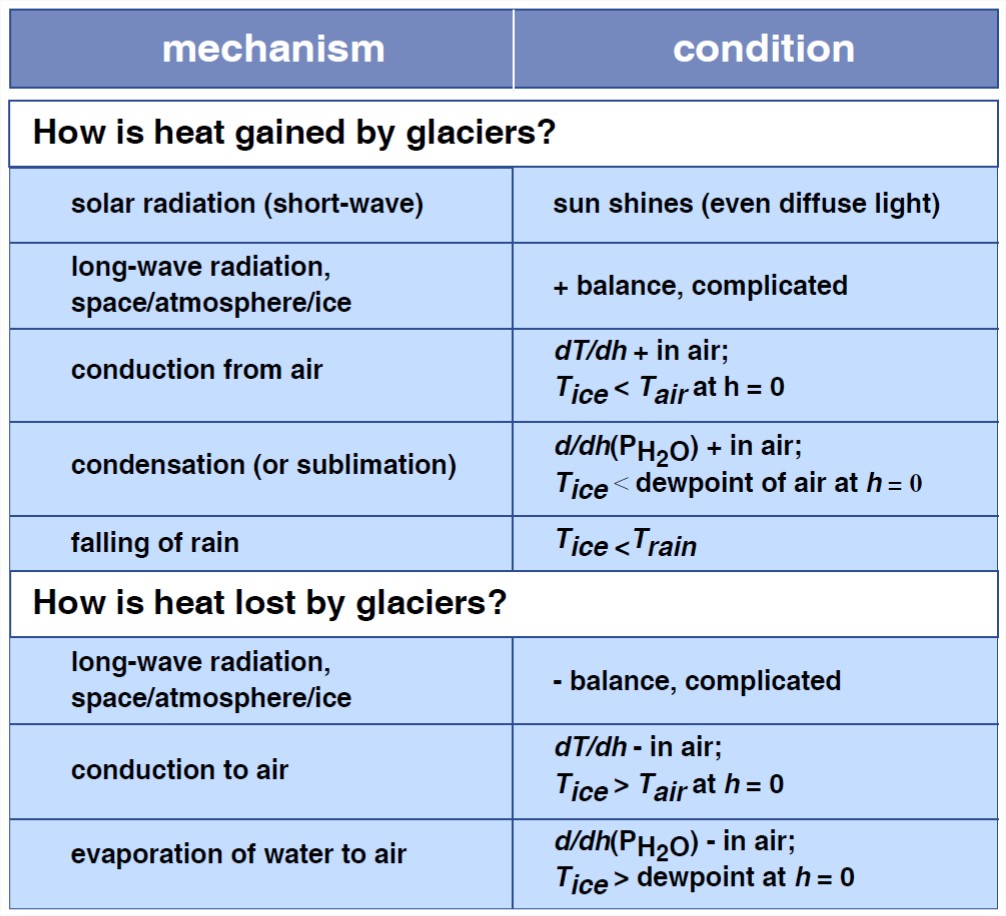
Table 7-1. Heat-exchange processes associated with a glacier.
An important indirect factor in all of this is heat conduction within the ice. This is the way heat is moved to or from the glacier surface, and it controls rates of radiation or conduction of heat at the glacier surface, because surface radiation and conduction is a function of the surface temperature of the ice.
Here are some miscellaneous descriptive points about Table 7-1, keyed by number:
(1) The solar constant is the rate at which the Sun delivers heat to the Earth. It’s conventionally taken to be the value that would be measured just outside the Earth’s atmosphere when the distance between the Sun and the Earth is at its mean annual distance. The value of the solar constant is very close to 2 cal/cm2-min.
(2) The albedo of the Earth is the percentage of incoming solar radiation that is reflected directly back to space, on the average. The albedo of a glacier varies considerably: snow surfaces have an albedo of 0.7–0.9, but glacier ice has an albedo of only 0.2–0.4.
(3) The surface energy exchange is greatly different under clear skies and under cloudy skies. Under clear skies, radiation directly to space is involved; typically the glacier loses heat, unless the air is very warm. Under cloudy skies, the direction of net flux of energy depends mostly on the relative temperature of clouds and ice.
(4) This would be minor without the effect of the wind. When the wind blows, there’s turbulent diffusion, which you know from the section on fluid dynamics in Chapter 1 to be much greater than molecular diffusion; then conduction to or from the overlying air can be very important.
(5) This is a minor effect.
(6) This isn’t important if the ice is at its melting point; 10 cm/day equals one day of long-wave radiation. But if the rain freezes when it falls, then this can be an important effect.
Energy exchange at the base of the glacier is a simpler matter, and the magnitudes of energy flux are not only much smaller but also far less variable. Geothermal heat is enough to melt about 5 mm of ice per year. This may not sound like much, but in a glacier whose basal ice is at the melting point it plays a significant role in glacier movement, by way of the lubricating effect of the thin film of water that’s continuously produced and then slowly drained away. There’s also heat from internal friction. This tends to be produced mainly in the lower part of the glacier, both because of basal slip and also because shearing is strongest in the lowermost part of the glacier. The rate of heat generation by internal deformation varies from considerably less to considerably more than the heat flux from the bedrock below, but it’s about the same order of magnitude.
Thermal Characteristics of Glaciers
The temperature characteristics of glaciers are important because deformation of ice, and therefore flow of glaciers, is strongly dependent on the temperature of the ice. At first thought it may seem that all glaciers are cold. But an important distinction can be made between
cold ice: ice below its pressure melting point (there can be no liquid water), and
warm ice: ice at the pressure melting point (there’s at least a little liquid water, under or between grains).
Recall from Chapter 1, in the section on water, that the melting point of ice falls slightly with increasing pressure. That translates to about a 2°C decrease beneath a thick ice sheet. Therefore there’s actually a downward temperature gradient in a glacier, although it’s small.
You can’t necessarily classify an entire glacier as warm or cold, because commonly the upper part or the geographical interior of the glacier has cold ice and the lower part or the lower-latitude fringes has warm ice. But in a given geographical region of the glacier, if the glacier is such that in winter all of the ice is below the melting point, and only the surficial part is raised to the melting point in summer, the glacier is said to be a cold glacier. On the other hand, if all of the ice is raised to the melting point in summer, and only a part is cooled below the melting point in winter, the glacier is said to be a warm glacier.
One of the most important consequences of the thermal structure of glaciers has to do with basal phenomena. Therefore we can talk about warm- based glaciers and cold-based glaciers. It’s generally believed that cold-based glaciers show little or no basal slip; the ice is frozen fast to the bedrock, and all movement is by internal deformation. On the other hand, a warm glacier has a thin layer of water at its base, facilitating basal sliding.
Cold ice is formed in two different ways:
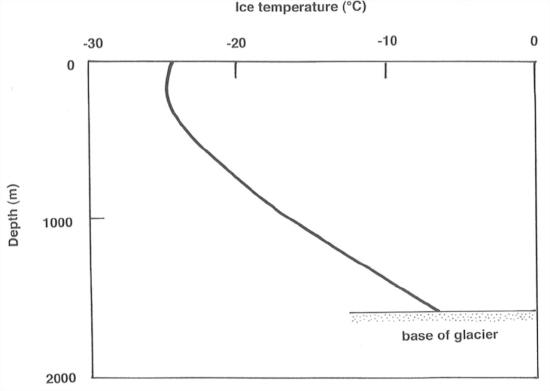
(1)Accumulationisatatemperaturesolowthatthere’snosurface melting during the summer. This is the case over most of the Antarctic ice sheet. The temperature of the firn and ice below the level of seasonal temperature change is approximately the same as the mean annual air temperature at the site. But temperature increases downward, because of geothermal heat. See Figure 7-26. Two opposing tendencies determine the course of the curve in Figure 7-26: (1) the value of geothermal heat flux from below, and (2) the rate of firn accumulation, which tends to "carry cold downward" into the ice sheet.
(2) Cooling the surface layer by winter cold. This effect extends down as much as 20 m. This happens at the surfaces of all glaciers in winter.
How is warm ice formed? By heating to raise the ice to its melting point (anywhere). This happens at the surface of the glacier, by one or more of the heat-transfer processes listed above, to form a surficial warm layer, and then meltwater percolates down and warms the ice by refreezing. This is an important effect: when 1 g of meltwater freezes, enough latent heat is released to raise 160 g of ice 1°C. Note that this warms the ice but doesn’t melt it. Note also that latent heat is the only source for warming at depth, because there’s almost no temperature gradient and therefore almost no conduction.
Warm ice is produced at the base of the glacier by basal heat sources. This is favored by the following circumstances:
• thick ice
• high surface temperature
• low accumulation rate
• high ice velocity
Over large areas of both the Antarctic and Greenland ice sheets, the basal ice is at its pressure melting point!
An important point is that when even a thin layer of warm ice is produced at the base of the glacier, the temperature gradient is about zero, so all the basal heat (frictional and geothermal) is used for melting, because there’s no conduction. This provides a continuous supply of meltwater at the base of the glacier.

