5.1: Distribution of Water on Earth
- Page ID
- 2257
Water is the second major earth system that we are discussing in depth. You will recall that geography is comprised of the ‘earth systems’ concept, in which the earth is made up of four systems: the atmosphere (which we discussed in week 3 and 4), the hydrosphere, the lithosphere and the biosphere. The hydrosphere is defined in your text as ‘an abiotic open system which includes all of the earth’s water’. Recall the definition of an ‘open system.’ It is a system with inputs and outputs which interact with surrounding systems. An abiotic system is a system which does not involve living organisms (in fact all earth systems except the biosphere are abiotic). Water is made up of two hydrogen and one oxygen atoms, and it has the familiar chemical formula H2O.
The hydrosphere contains about 1.36 billion cubic kilometers of water, and 71% of the earth’s surface is covered with water. Conduct some research and fill in the appropriate distribution of the earth’s hydrosphere:
Ocean________
All fresh water________
o Groundwater________
o Deep groundwater________
o Soil moisture________
o Surface________
- Ice and glaciers________
- Freshwater lakes________
- Rivers and streams________
- Saline lakes________
- Atmosphere________
The water cycle is an open system. Water is lost from the system when gaseous water in the atmosphere disassociates (breaks apart) when it is hit by high energy waves from the sun (insolation). Because hydrogen is the lightest element, it often will escape the earth’s gravitational pull and be lost to space. Water is gained by the system by ‘outgassing’ from the earth’s crust. When the liquid rock in the interior of the earth’s crust rises to the surface and cools, certain compounds precipitate out, including water. This water is very hot, so it is in its gaseous form (steam), and it can often be seen rising from geothermal (a term that means hot earth) areas.
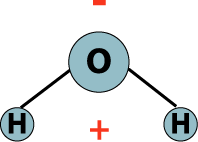
Water is a polar molecule (see the model of it above), meaning that one side of the molecule has a positive charge, and the other side of the molecule has a negative charge. This polarity is the reason behind the many unique properties of water, because the positive and negative ends of water stick to each other in a loose form of bonding called ‘hydrogen bonding’. Hydrogen bonding is the reason that liquid water is ‘wet’ because it sticks to many things and dissolves many substances. Hydrogen bonding also is the cause of capillary action of ‘wicking’ where water will climb up a narrow tube or space (such as between the fibers of a towel). Capillary action is the way that most plants are able to get water from their roots to their leaves. Hydrogen bonding is also the reason that solid water (ice) arranges itself into ring-shaped structures which actually take up more space than liquid. This is why water is the only substance that expands as it freezes, making it a powerful and destructive force on our planet.


